Notes 21 - CEProfs
advertisement

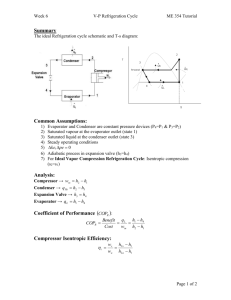
Lec 21: Isentropic efficiencies, air standard cycle, Carnot cycle, Otto cycle 1 • For next time: – Read: § 8-6 to 8-7 – HW11 due Wednesday, November 12, 2003 • Outline: – Isentropic efficiency – Air standard cycle – Otto cycle • Important points: – Realize that we already know how to analyze all these new cycles, we just need to define what the cycle steps are – Know the difference between the air standard cycle and the cold air approximations – Know how to solve cycles using variable specific heats and constant specific heats 2 Isentropic Efficiencies We can use the isentropic process as an ideal by which to compare real processes in different engineering devices. COMPRESSORS Ws ws c Wact w act Actual compressors take more work than isentropic compressors. The efficiency will vary between zero and one. 3 Compressor Isentropic Efficiency For a steady-state, adiabatic compressor q w h 2 h1 KE PE If q, KE, and PE are all zero, then: w h1 h 2 General expression w s h 1 h 2s Isentropic compressor Actual compressor w a h1 h 2a 4 Compressor Isentropic Efficiency The compressor efficiency is then: w s h1 h 2s c w a h1 h 2a For an ideal gas with constant specific heats, w s h 1 h 2 s c p ( T1 T2 s ) w a h 1 h 2a c p (T1 T2a ) Thus: T1 T2s c T1 T2a 5 Compressor Isentropic Efficiency Note that the work is directly proportional to T with constant specific heats. Real gases will also have dependence on P. The work can then be represented by a change on the T-axis of a Ts diagram. 6 Compressor Isentropic Efficiencies T 2a 2s T1–T2a T1–T2s 1 S 7 Isentropic Efficiencies TURBINES With turbines, we’re interested in the work/power output not input. An isentropic turbine will produce the maximum output. Efficiency is given by: Wact w act T Ws ws h1 h 2a T h1 h 2s Again, isentropic efficiency will vary between zero and one. 8 Isentropic Efficiencies T 1 2a 2s 2a 2a S Actual path will vary depending on amount of irreversibilities ... 9 Isentropic Efficiencies For nozzles, the isentropic efficiency is given by Vexit 2 a N 2 Vexit 2 s 2 10 TEAMPLAY • Work problem 7-89 11 Chapter 8, Gas Cycles • Carnot cycle is the most efficient cycle that can be executed between a heat source and a heat sink. TL 1TH • However, isothermal heat transfer is difficult to obtain in reality--requires large heat exchangers and a lot of time. 12 Gas Cycles • Therefore, the very important (reversible) Carnot cycle, composed of two reversible isothermal processes and two reversible adiabatic processes, is never realized as a practical matter. • Its real value is as a standard of comparison for all other cycles. 13 Gas Cycles • Assumptions of air standard cycle • Analyze two cycles in detail – Otto – Brayton 14 Assumptions of air standard cycle • Working fluid is air • Air is ideal gas • Combustion process is replaced by heat addition process • Heat rejection is used to restore the fluid to its initial state and complete the cycle • All processes are internally reversible • Constant or variable specific heats can be used 15 Gas cycles have many engineering applications • Internal combustion engine – Otto cycle – Diesel cycle • Gas turbines – Brayton cycle • Refrigeration – Reversed Brayton cycle 16 Some nomenclature before starting internal combustion engine cycles 17 More terminology 18 Terminology • • • • • Bore = d Stroke = s d 2 Displacement volume =DV = s 4 Clearance volume = CV Compression ratio = r VBDC DV CV r CV VTDC 19 Mean Effective Pressure Mean Effective Pressure (MEP) is a fictitious pressure, such that if it acted on the piston during the entire power stroke, it would produce the same amount of net work. Wnet MEP Vmax Vmin 20 The net work output of a cycle is equivalent to the product of the mean effect pressure and the displacement volume 21 Real Otto cycle 22 Real and Idealized Cycle 23 Idealized Otto cycle 24 Idealized Otto cycle • 1-2 - ADIABATIC COMPRESSION (ISENTROPIC) • 2-3 - CONSTANT VOLUME HEAT ADDITION • 3-4 - ADIABATIC EXPANSION (ISENTROPIC) • 4-1 - CONSTANT VOLUME HEAT REJECTION 25 Performance of cycle Efficiency: w net q in Let’s start by getting heat input: q in u 3 u 2 26 Cycle Performance Get net work from energy balance of cycle: net in out q q w Substituting for qin and qout: w net (u 3 u 2 ) (u 4 u1 ) Efficiency is then: w net q in 27 Cycle Performance Substituting for net work and heat input: (u 3 - u 2 ) - (u 4 - u1 ) (u 3 - u 2 ) We can simplify the above expression: (u 4 - u1 ) 1 (u 3 - u 2 ) 28 Teamplay Problem 8-36 29 Cold air standard cycle cp, cv, and k are constant at ambient temperature ( 70 °F) values. Assumption will allow us to get a quick “first cut”approximation of performance of cycle. 30 Cycle performance with cold air cycle assumptions If we assume constant specific heats: c v (T4 - T1 ) (u 4 - u1 ) 1 1 (u 3 - u 2 ) c v (T3 - T2 ) (T4 - T1 ) 1 (T3 - T2 ) 31 Cycle performance with cold air cycle assumptions Because we’ve got two isentropic processes in the cycle, T1 can be related to T2, and T3 can be related to T4 with our ideal gas isentropic relationships…. Details are in the book! T2 V1 T1 V2 k 1 r k 1 T4 V4 T3 V3 T4 T3 Thus T1 T2 k 1 1 r k 1 32 Cycle performance with cold air cycle assumptions T1 1 1 1 k 1 T2 r This looks like the Carnot efficiency, but it is not! T1 and T2 are not constant. What are the limitations for this expression? 33 Differences between Otto and Carnot cycles T 2 33 2 44 1 3 s 34 Effect of compression ratio on Otto cycle efficiency 35 Sample Problem The air at the beginning of the compression stroke of an air-standard Otto cycle is at 95 kPa and 22C and the cylinder volume is 5600 cm3. The compression ratio is 9 and 8.6 kJ are added during the heat addition process. Calculate: (a) the temperature and pressure after the compression and heat addition process (b) the thermal efficiency of the cycle Use cold air cycle assumptions. 36 Draw cycle and label points 3 r = V1 /V2 = V4 /V3 = 9 P Q23 = 8.6 kJ 2 4 T1 = 299 K 1 P = 95 kPa 1 v 37 Major assumptions • • • • Kinetic and potential energies are zero Closed system 1 is start of compression Ideal cycle: 1-2 isentropic compression, 2-3 const. volume heat addition, etc. • Cold cycle const. properties 38 Carry through with solution Calculate mass of air: P1V1 m 6.29 x 10-3 kg RT1 Compression occurs from 1 to 2: V1 T2 T1 V2 k 1 isentropic compressio n T2 22 273K 9 1.4 1 T2 705.6 K But we need T3! 39 Get T3 with first law: Q23 W m(u ke pe) mc v T3 T2 Solve for T3: q 8.6 kJ 6.29x10 3 kg 705.6 K T3 T2 cv 0.855 kJ kg T3 2304.7 K 40 Thermal Efficiency 1 1 r k 1 1 1 1.4 1 9 0.585 41 Let’s take a look at the Diesel cycle. 42 Idealized Diesel cycle 1-2 - ADIABATIC COMPRESSION (ISENTROPIC) 2-3 - CONSTANT PRESSURE HEAT ADDITION 3-4 - ADIABATIC EXPANSION (ISENTROPIC) 4-1 - CONSTANT VOLUME HEAT REJECTION 43 Performance of cycle Efficiency: w net q in Heat input occurs from 2 to 3 in constant pressure process: q in h 3 h 2 Why enthalpies? 44 TEAMPLAY • Work problem 8-16 45