Environmental chemistry air pollution
advertisement

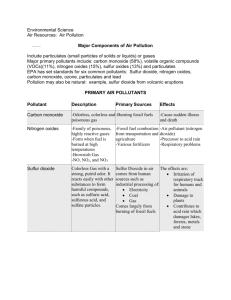
Environmental chemistry study of the effect of human activity on the chemical processes in the environment concerns political and natural borders global issues applied chemistry Main topics: core air pollution acid deposition greenhouse effect ozone depletion dissolved oxygen in water water treatment soil waste Main topics: AHL ozone depletion smog acid deposition water and soil Environmental chemistry E1: air pollution Describe the main sources of carbon monoxide (CO), oxides of nitrogen (NOx), oxides of sulfur (SOx), particulates and volatile organic compounds (VOCs) in the atmosphere. Evaluate current methods for the reduction of air pollution. the atmosphere composition of dry clean air http://www.dep.state.pa.us/earthdaycentral/97/air_teachers/TAB1.htm component content component % by Vol. ppm Nitrogen 78.09 780,900 Oxygen 20.94 Argon content % by Vol. ppm Hydrogen .00005 0.5 209,400 Methane .00015 1.5 .93 9,300 Nitrogen dioxide .0000001 0.001 Carbon dioxide .0318 318 Ozone .000002 0.02 Neon .0018 18 Sulfur dioxide .00000002 0.0002 Helium .00052 5.2 Carbon monoxide .00001 0.1 Krypton .0001 1 Ammonia .000001 0.01 Xeon .000008 0.08 Nitrous oxide .000025 Units used to express amount of a gas % by volume ppm = parts per million/particles per million particles Remember in gases, assuming conditions are the same: same volume = same number of particles So 78.09 % by volume is 78.09 % particles per million particles or 780,900 ppm primary air pollutants waste products from human activity primary = added directly to the air pollutant = chemical in the wrong concentration in the wrong place primary air pollutants: CO NOx particulates SOx volatile organic compounds (VOCs) Air pollutants For each air pollutant you need to know: sources: natural and man-made methods of reducing its emissions any relevant balanced symbol equations carbon monoxide: sources Natural: atmospheric oxidation of methane CH4 + ½ O2 CO + 2H2 natural forest fires Man-made: incomplete combustion of carbon-containing fuels; mainly in cities; localized problem as well as fluctuations during the day – heavy traffic. forest fires carbon monoxide: health effect CO combines with Fe in haemoglobin in blood – bonds 320 times stronger than oxygen – oxygen cannot bond onto heamoglobin. Less oxygen supplied to body cells. Effects: headaches, shortness of breath, in case of high concentration (e.g. rush hour): unconsciousness, death. carbon monoxide: reduction (1) Lean burn engines Decrease fuel /air ratio (in mass) from 1:14.7 (stoichiometric ratio/green band in diagram) to 1:18. Why? (or increase air /fuel ratio to 18:1) carbon monoxide: reduction (2) catalytic converter in car exhaust system reduction of nitrogen oxide oxidation of CO and unburnt hydrocarbons/VOCs 2CO (g) + O2 (g) 2CO2 (g) 2NO (g) + 2CO (g) 2CO2 (g) + N2 (g) 2C8H18 (g) + 25O2 (g) 16CO2 (g) + 18H2O (g) Catalytic converter catalytic converter To achieve high rate of reaction for this redox process: exhaust gases are passed over platinum which acts as a heterogeneous catalyst; converter has a honey comb structure to increase surface area; very high temperature of the exhaust gases also increase the rate of reaction dramatically. Thermal exhaust reactor Heat from engine makes CO react with more air to oxidise it further to carbon dioxide. sulphur oxides: sources Natural: volcanoes sea spray biological decay of organic matter that contains sulphur reduction of sulphates Man-made: coal-burning power stations (equation starting from S in coal) roasting of metal sulphides e.g. ZnS and Cu2S (equation) sulphur oxides: health effects acidic oxides lung irritants, affect in particular those suffering from respiratory problems e.g. asthma sufferers formation of sulphuric acid aerosols (droplets of sulphuric acid) (equation) (often catalysed by metal particulates); effects of aerosols: irritant to the eyes irritate vessels in lungs causing impaired breathing sulphur oxides: methods of reduction use of “low-sulphur content” fuels or removal of sulphur before burning coal/oil removal of SO2 from fumes before they are released: limestone based fluidised bed (equations showing decomposition of calcium CaCO3 reaction of CaO with SO2) alkaline scrubbing (wet scrubber) (equations showing reaction of CaO and Mg(OH)2) Sulphur oxides: reduction (1) Wet scrubber Limestone based fluidised bed nitrogen oxides: sources Natural: Electrical storms release enough energy to cause oxidation of atmospheric nitrogen: (equations showing oxidation of nitrogen and further oxidation of NO) Decomposition of organic matter containing nitrogen Man-made: Combustion of fossil fuels in car engines and furnaces of power stations produces high temperatures to cause oxidation : (equation showing oxidation of nitrogen) nitrogen oxides: health effects choking irritating gas, affects eyes and people with respiratory problems forms nitric acid aerosols/acid rain (equation showing dissolution of nitric acid in water) nitric acid also increases the rate of oxidation of SO2 (see later) plays an important role in the formation of secondary pollutants e.g. ozone and smog nitrogen oxides: reduction catalytic converter lean burn engines: high air/fuel ratio or low fuel/air recirculation/reburn of exhaust gases: nitrogen oxide emissions are reduced by reintroducing exhaust gases into the fuel mixture, lowering peak combustion temperatures as it is the high temperature in the combustion engine which causes nitrogen oxide production. Particulates: sources particulates = airborne/suspended liquid and solid particles. Natural: volcanic eruptions large forest fires Man-made: burning fossil fuels e.g. diesel forest fires industrial emissions; chemical processes incinerators Particulates: health effects particulates penetrate lungs and may block air passages some are poisonous e.g. Pb and asbestos adsorb chemicals and can act as catalysts in reactions producing secondary pollutants by adsorbing also increase concentration and rate of reaction reduce visibility Particulates: reduction Electrostatic precipitator . Particulates are given a negative charge and then attracted onto positively collection plates Particulates: reduction Volatile organic compounds: sources Natural sources: methane: bacterial anaerobic decomposition of organic matter (e.g. in rice paddies) from plants e.g. terpenes leakage from natural fossil reserves Man-made: evaporation of fuels partial combustion of fuels leakage from storage reservoirs VOCs: health effects photochemical smog can lead to carcinogenic compounds fatigue, weakness respiratory problems VOCs: reduction catalytic converter