Group 16. Sulfur

Group 15 Nitrogen
(Section 22.7, p.962)
Nitrogen, N
2
, - diatomic gas containing N
N triple bond.
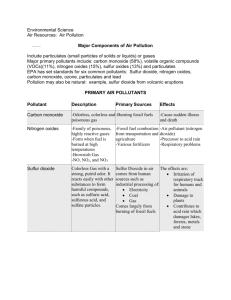
Nitrogen compounds show wide range of
O.S.’s and coordination numbers (C.N.),
N
2
O, NO, NH
3
, NH
4
Cl, NaNO
3
.
Note: C.N. of an element in a covalent compound is defined as the no. of other atoms or groups covalently bonded to it. e.g. C.N. of N in NH
3
is 3; C.N. of N in
NaNO
3
is also 3.
One of the most important compounds of nitrogen is ammonia, NH
3
. Industrially made by HABER process:
N
2
+ 3H
2
2NH
3
Several nitrogen oxides known since nitrogen can have variety of O.S.’s, e.g. N
2
O, NO and NO
2
. Different reaction conditions give different oxides.
Note: O always has O.S. –2 in these compounds (Why?)
N oxides are covalent oxides, hence react with water giving acidic solutions (section
3.6). For example:
Reaction of nitrogen dioxide and water gives the products nitric acid and nitrogen monoxide.
Question:
Write the balanced equation for this reaction.
NO
2
+ H
2
O
HNO
3
+ NO (Not balanced!)
Give the O.S. of each element in this equation and hence say whether this is a redox reaction.
You use nitric acid in the practical course. It is a strong acid and also a strong oxidising agent i.e. it oxidises compounds and is itself reduced. e.g.
C + 4HNO
3
CO
2
+ 4NO
2
+2H
2
O
When nitric acid reacts with a base, (acidbase reaction), the products are the nitrate salt and water. e.g.
2HNO
3
+ Ca(OH)
2
Ca(NO
3
)
2
+ 2H
2
O
(acid) (base) (salt) (water)
Nitrates metabolised by most plants so useful as fertilisers.
Group 15 Phosphorus
(Section 22.8, p.967)
P exists as a molecular compound, P
4
, with the P atoms arranged in a tetrahedral fashion.
P
P P
P
PH
3
(the P analogue of NH
3
) is known. Is a toxic, colourless gas which smells of fish.
Cannot be formed by direct reaction between P
4
and H
2
. Possible reason: P & H have same electronegativity value (2.1).
Instead to make PH
3
, react an acid with an ionic phosphide. e.g. Na
3
P + 3HCl
3NaCl + PH
3
(base) (acid)
P
4
reacts with oxygen to form phosphorus oxides. Unlike nitrogen, only two oxides are stable - P
4
O
6
and P
4
O
10
.
Formed by direct reaction of P
4
and O
2
.
Nature of product depends on the stoichiometry (ratio) of the reactants.
P
4
+ 3O
2
P
4
O
6
(O.S. of P is +3)
P
4
+ 5O
2
P
4
O
10
(O.S. of P is +5)
Structures:
O
O
P
O
P
O O
P
O
P
O
O
O
P
O
P
O
O O
P
P
O
(3-D structures on CD, ch. 22 )
O
O
P oxides are covalent oxides and so react with water giving acidic solutions (section
3.6).
Either
P
4
O
6
+ 6H
2
O
4H
3
PO
3
H
3
PO
3
is phosphor ous acid. O.S.(P) +3.
Or
P
4
O
10
+ 6H
2
O
4H
3
PO
4
H
3
PO
4
is phosphor ic acid. O.S. (P) +5.
Phosphoric acid used as a preservative, e.g. in soft drinks (Coke), detergents, pharmaceuticals etc.
Note: H
3
PO
3
and H
3
PO
4
are oxy acids of phosphorus.
P
4
reacts with Group 17 (the halogens) to give compounds of the type PX
3
and PX
5
(X
= halogen, i.e. F, Cl, Br). Product obtained depends on ratio of reactants.
P
4
+ 6Cl
2
4PCl
3
P
4
+ 10Cl
2
4PCl
5 i.e. Cl
2
will oxidise P to +3 oxidation state if low ratio of Cl
2
present, and to +5 oxidation state if high ratio Cl
2
present.
PCl
3
and PCl
5
undergo hydrolysis reactions to form oxyacids.
PCl
3
+ 3H
2
O
3HCl + H
3
PO
3
PCl
5
+ 4H
2
O
5HCl + H
3
PO
4
Group 16.
Sulfur
(Section 22.6, p.958)
Sulfur – exists as a molecular compound, with the atoms arranged in a crown fashion.
S
S
S
S
S S
S
S
(3-D model on CD, ch. 22 )
H
2
S (the S analogue of H
2
O) is known. Best made by reaction of an ionic sulfide with acid, e.g.
2HCl + Na
2
S
2NaCl + H
2
S
(acid) (base)
Note: Cannot make H
2
S by direct reaction of
S
8
with H
2
; these elements do not react readily together.
Sulfur oxides:
2 common oxides
(a) SO
2
(sulfur dioxide) made by combination reaction:
S
8
+ 8O
2
8SO
2
SO
2
used to sterilise dried fruit and wine as it is toxic to microorganisms.
(b) SO
3
(sulfur trioxide) made by combination of SO
2
and O
2
. Reaction is slow, so need catalyst, e.g. V
2
O
5
(an oxide of the d-block element, vanadium) to speed it up.
2SO
2
+ O
V
2
O
5
2
2SO
3
Catalyst –substance that changes rate of reaction but is itself not used up in reaction.
S oxides are covalent oxides and so react with water giving acidic solutions.
SO
2
+ H
2
O
H
2
SO
3
H
2
SO
3
is sulfur ous acid. S in O.S. +4.
SO
3
+ H
2
O
H
2
SO
4
H
2
SO
4
is sulfur ic acid. S in O.S. +6.
Sulfuric acid - strong acid and moderately good oxidising agent.