Periodic Table - jamietucker13
advertisement

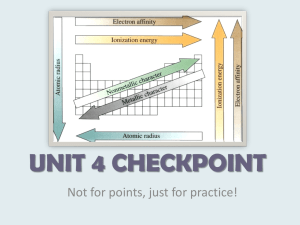
Hydrogen is a NONMETAL despite its placement on the left hand side of the P.Table. Metals-Left of “staircase” Nonmetals-Right of “staircase” Alkali metals-first family Alkaline Earth metals-second family Halogens-second to last family Noble gases-last family Lanthanides-pulled out first row Actinides-pulled out second row Group 1 (excluding Hydrogen) React with water violently! Lose a valence electron to become 1+ Cations Soft, highly reactive, good conductors of heat and electricity, Li › Lithium batteries, lithium in dehumidifiers, strengthens glass, used to treat mood swings, fireworks. Na › Found in table salt, vapor lamps, sodium potassium pump in animals to create gradient in cells, fireworks. K › Active transport pump, nerve cell conduction, fertilizer, salt substitute, fireworks. Group 2 Shiny solids, harder than alkali metals Lose 2 valence electrons, 2+ charge Cations React with water (except Beryllium) React with oxygen Calcium› bones, teeth, bloodstream, limestone, chalk, marble, coral reef, Magnesium› alloys of magnesium and aluminum and zinc are strong, but light › Plants need it to make chlorophyll › Found in hard water (makes it difficult for soap to work properly › Strontium-fireworks-red color Group 17 (sometimes referred to as 7A) Forms compounds with almost all metals. Gain one valence electron to become 1- charge Anions Fluorine › Toothpaste, drinking water Chlorine › Gas at room temperature, deadly gas, bleaching agents, anesthetics at the dentist, disinfects water, found in stomach acid. Bromine › Used in photographic film Iodine › Need in diet to maintain healthy thyroid gland, kills bacteria Group 18 (sometimes listed as 8A) Last naturally occurring elements to be discovered. Xe will react with F (F is so reactive) Fairly inert (stable) Gas at room temperature He› used to inflate balloons, sun, deep sea diving tanks Ne› light displays (electricity excites electrons, when they fall it releases energy in the form of light) Ar› most abundant noble gas on Earth, atmosphere, welding, Left of staircase Cations Give up electrons Good conductors of heat and electricity High melting and boiling points. Generally solids, at room temperature. Malleable (sheets) and Ductile (wire) Either side of the staircase Physical and chemical properties of both metals and nonmetals. Used in computer chips and solar cells Germanium and Silicon are the most used. B, Si, Ge, As, Sb, Te, Po, Upper right side of periodic table (right of staircase) Generally gases, Generally brittle and dull Poor conductors of heat and electricity The only liquid at room temperature is Bromine, others are all gases 1-18 as you go Across in the periodic table. The number of electrons occupying the outermost energy level. 1-8, s and p blocks. 1-8 main group elements. Each family has the SAME number of valence electrons. s, p, d, f The group with one valence electron and very reactive with water is the….. Alkali metals The block that contains alkali metals and alkaline earth metals. S block The elements that are ON the staircase are referred to as……..because they have both properties of metals and nonmetals. metalloids Fluorine is in the family….. halogens Group 13 contains how many valence electrons? 3 Group 18 contains how many valence electrons? 8 except for Helium which only contains 2 These elements are good conductors of heat and electricity and make up most of the elements on the periodic table. metals These are mostly gases at room temperature except Bromine which is a liquid at room temperature. nonmetals At atom that loses an electron is termed a…. cation An atom that gains an electron is termed an…. anion