Gas Laws
advertisement

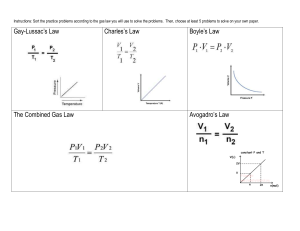
In This Lesson: Gas Laws (Lesson 3 of 3) Stuff You Need: Calculator Today is Tuesday, December 22nd, 2015 Pre-Class: Did you take a worksheet? Today’s Agenda • Balloon Demo • Gas Laws – Got gas? • Computer Tutorial • Uh…more gas laws? • Oh, and I’ll unravel one of life’s mysteries. • Where is this in my book? – P. 413 and following… By the end of this lesson… • You should be able to identify the volume, temperature, pressure, or quantity of a sample of gas. • You should be able to describe several gas laws developed to make such calculations. Aside: Jan van Helmont • Belgian physician – tries to figure out if plants take mass from soil to grow so large. • Measured soil, planted a seed, and watered it. • Then he waited five years (!). The soil weighed the same as it did five years earlier. • CONCLUSION – The plant is using water (hydrate) to grow. (it’s also using CO2 but he didn’t catch that part) – Jan van Helmont also coined the word “gas.” http://2.bp.blogspot.com/_XU9nEwykZRc/SkO5s53xzgI/AAAAAAAAACU/gUPGWU3wpbo/s320/van_Helmont.jpg Floating head! Spooky… Gases • Unlike solids and liquids, gases can be compressed easily. – Imagine squeezing a balloon without popping it. – Now try to imagine squeezing the water in a cup, or squeezing an ice cube. Not easily done. • The reason for this is that the atoms or molecules in a gas are relatively far apart from one another. Particles in Each Phase http://www.quantumtheatre.co.uk/BigDrip%20ks1_files/image008.jpg Balloon Demo • In addition to compressibility, gases are also dramatically affected by temperature. • It’s these two things I can exploit to perform this demo. • Balloon in a flask! – Why does a rapidly chilled flask with a balloon on top get “sucked” the flask? – Because the outside air pressure is greater and forces it inside. It’s actually getting pushed in. A Quick Reminder • Recall that one mole of a gas…any gas…takes up 22.4 L of volume. – One mole of H2 has a molar mass of approximately 2 g/mol. It takes up 22.4 Liters. – One mole of CH4 has a molar mass of approximately 16 g/mol. It takes up 22.4 Liters. • Also: • 1 atm = 101.3 kPa = 760 torr = 760 mm Hg An Introduction to Variables • Gas laws and equations use a lot of variables. Here are some: – P = Pressure – V = Volume – n = Number of Moles – T = Temperature (Kelvin) • Another introduction to variables: – Gas Laws Tutorial (computers) Under Pressure • Pressure, you may remember, is caused by collisions of molecules with each other and the walls of the container. • Therefore, pressure is expressed in terms of force per unit area. • Think of it like walking on very deep snow. – Your weight is always the same, but depending on how that weight is distributed, some shoes will be better choices than others. Standard Pressure • • • • • 1 atm 101.3 kPa 760 mm Hg 760 torr 14.7 psi (lbs/in2) P is for Pressure • Pressure increases if: – Amount of gas increases. – Temperature increases. – Volume decreases. • Pressure decreases if: – Amount of gas decreases. – Temperature decreases. – Volume increases. P is for Pressure • Pressure increases proportionately with amount of gas molecules. – Imagine a container with constant volume and 100 particles inside. – You add 100 more particles. • The original pressure has doubled. – You add 100 more particles. • The original pressure has tripled. • Now imagine the pressure exceeds the strength of the container. – Pop! P is for Pressure • The air in soda bottles is under high pressure. What happens when you open the cap? – Air rushes out because P is lower outside. • Sometimes jars of nuts are vacuum packed. What happens when you open them? – Air rushes in because P is higher outside. • In other words, gases move from areas of high pressure to areas of low pressure until pressures equalize at equilibrium. Pressure Example: Spray Cans • Gas inside is at high pressure; volume is constant. • Inside gas tries to expand itself, and in doing so pushes out paint. Aside: Air Pressure • During weather reports you’ll frequently hear about “low pressure areas” or “high pressure zones.” – These are, quite literally, areas of lower or higher atmospheric pressure, somewhat due to solar heating. • Low pressure air lets water evaporate and ascend more easily, thus being associated with rain. • Also, because our atmosphere “wants” to be at a constant pressure all the time, air will move from high pressure areas to low pressure areas. • The result of that movement? – Wind! V is for Volume • Volume is the space taken up by matter. • Increasing volume: – Decreases pressure. – Decreases temperature. • Decreasing volume: – Increases pressure. – Increases temperature. V is for Volume • Imagine a 1 L container. • You cut the volume in half. • What happens to the pressure? – It doubles! • Thus, pressure and volume are inversely proportional to one another. – Volume down, pressure up. – Volume up, pressure down. T is for Temperature • Temperature is a measure of average kinetic energy. • Temperature increases if: – Pressure increases. – Volume decreases. • Temperature decreases if: – Pressure decreases. – Volume increases. T is for Temperature • Those of you with cars may know that it’s not a good idea to check tire pressure after driving. Why? – Friction with the road causes the tires to get hotter. – Increased temperature causes pressure to increase, providing an inaccurate reading. • This is the same reason balloons that spend a long time on lights tend to pop. Temperature Scales Celsius and Kelvin • Remember that to convert Celsius to Kelvin (and vice versa), use the following formulas: • °C = K – 273 • K = °C + 273 • Also recall that STP (standard temperature and pressure) is equivalent to: • 273 K or 0 °C • 1 atm / 101.3 kPa / 760 mm Hg / 760 torr / 14.7 psi Quick Note • Be careful with the wording when it comes to relationships between variables. • For example, we learned that a decrease in volume causes a increase in temperature. • However, if I were to increase the temperature of a sample of gas, the volume would also increase. • The key is to understand what’s happening at a molecular level. – Think about particle collisions, mainly. Gas Laws • So, with all these pressure/volume/temperature relationships, you can probably imagine that chemists had a field day. • Here’s what they came up with: Boyle’s Law • Proposed by Robert Boyle, 1662: • Pressure is inversely proportional to volume when temperature is held constant: P1V1 P2V2 • • • • P1 is like “pressure before.” V1 is like “volume before.” P2 is like “pressure after.” V2 is like “volume after.” – This whole “before/after” thing applies in nearly all the rules. Boyle’s Law Graph Boyle’s Law Practice • 20.5 L of nitrogen at 25 °C and 742 torr are compressed to 9.8 atm at constant T. What is the new volume? – Before: 20.5 L, 742 torr – After: 9.8 atm • Let’s convert atm to torr first. • 9.8 atm = 7448 torr • P1V1 = P2V2 • 742 torr * 20.5 L = 7448 torr * V2 • V2 = 2.04 L Boyle’s Law Practice • 30.6 mL of carbon dioxide at 740 torr is expanded at constant temperature to 750 mL. What is the final pressure in kPa? – Before: 30.6 mL, 740 torr – After: 750 mL • P1V1 = P2V2 • 740 torr * 30.6 mL = P2 * 750 mL • P2 = 30.19 torr (must be expressed in kPa) • P2 = 4.024 kPa Boyle’s Law Practice • Gas Laws Packet – Boyle’s Law page, odd numbers and #6 Charles’ Law • Proposed by Jacques Charles, 1787: • The volume of a gas is directly proportional to temperature when pressure is held constant (volume is zero at 0 K). • Temperature must be in Kelvin! V1 V2 T1 T2 Charles’ Law Graph Charles’ Law Practice • What would the final volume be if 247 mL of gas at 22 °C is heated to 98 °C , if the pressure is held constant? – Before: 247 mL, 22 °C (295 K) – After: 98 °C (371 K) • V1/T1 = V2/T2 • 247 / 295 = V2 / 371 • V2 = 310.63 mL Charles’ Law Practice • At what temperature would 40.5 L of gas at 23.4 °C have a volume of 81.0 L at constant pressure? – Before: 40.5 L, 23.4 °C (296.4 K) – After: 81.0 L • V1/T1 = V2/T2 • 40.5 / 296.4 = 81.0 / T2 • Cross-multiply! • T2 = 592.8 K = 319.8 °C Charles’ Law Practice • Gas Laws Packet – Charles’ Law page, odd numbers and #2 Gay-Lussac’s Law • Proposed by Joseph Gay-Lussac, 1802: • The pressure and temperature of a gas are directly related when volume is held constant. • Temperature must be in Kelvin! P1 P2 T1 T2 Gay-Lussac’s Law Graph Gay-Lussac’s Law Practice • A sample of nitrogen gas has a pressure of 6.58 kPa at 539 K. If the volume does not change, what will the pressure be at 211 K? – Before: 6.58 kPa, 539 K – After: 211 K • P1 / T1 = P2 / T2 • 6.58 / 539 = P2 / 211 • P2 = 2.58 kPa Gay-Lussac’s Law Practice • The pressure in a car tire is 198 kPa at 27 °C. After a long drive, the pressure is 225 kPa. What is the temperature of the air in the tire? Assume that the volume is constant. – Before: 198 kPa, 27 °C (300 K) – After: 225 kPa • P1 / T1 = P2 / T2 • 198 / 300 = 225 / T2 • Cross-multiply! • T2 = 340.9 K = 67.9 °C Gay-Lussac’s Law Practice • Gas Laws Packet – Gay-Lussac’s Law page, odd numbers Avogadro’s Law/Hypothesis • Proposed by Amedeo Avogadro, 1811: • Hypothesis: Two equal volumes of gas, at the same temperature and pressure, contain the same number of molecules. • Law: The volume and number of moles of a gas are directly related when temperature is held constant. V1 V2 n1 n 2 Combined Gas Law • The combined gas law expresses the relationship between pressure, volume and temperature of a fixed amount of gas. • Temperature must be in Kelvin. P1V1 P2V2 T1 T2 Combined Gas Law Practice • A deodorant can has a volume of 175 mL and a pressure of 3.8 atm at 22 °C. What volume of gas could the can release at 22 °C and 743 torr? – Before: 175 mL, 3.8 atm, 22 °C (295 K) – After: 743 torr, 22 °C (295 K) • P1V1 / T1 = P2V2 / T2 • 3.8 atm = 2888 torr • (2888 * 175) / 295 = (743 * V2) / 295 • V2 = 680.21 mL Combined Gas Law Practice • Gas Laws Packet – Combined Gas Law page, odd numbers Summary of Laws • Boyle’s Law • P1V1 = P2V2 • Charles’ Law • V1 / T1 = V2 / T2 • Gay-Lussac’s Law • P1 / T1 = P2 / T2 • Avogadro’s Law/Hypothesis • V1/n1 = V2/n2 • Combined Gas Law • P1V1 / T1 = P2V2 / T2 Ideal Gases • Ideal gases are imaginary gases that perfectly fit all of the assumptions of the kinetic molecular theory: – Gases consist of tiny particles that are far apart relative to their size. – Collisions between gas particles and between particles and the walls of the container are elastic collisions. – No kinetic energy is lost in elastic collisions. Ideal Gases (Continued) • Gas particles are in constant, rapid motion. They therefore possess kinetic energy, the energy of motion. • There are no forces of attraction between gas particles. • The average kinetic energy of gas particles depends on temperature, not on the identity of the particle. Real Gases • Real gases do not behave ideally: – Real gases do experience intermolecular attraction. – Real gases do have volume. – Real gases do not have elastic collisions. • Large, polar gases at low temperature and high pressure tend not to behave ideally. • Small, non-polar gases at high temperature and low pressure tend to behave almost ideally. Real Gases Video • CrashCourse – Real Gases Ideal Gas Law • • • • • • PV = nRT P – pressure (in kPa or atm or mm Hg/torr) V – volume (L) n – moles T – temperature (K) R – Ideal Gas Constant – depends on pressure unit: • R = 8.314 LkPa / Kmol • R = 0.0821 Latm / Kmol • R = 62.38 Ltorr / Kmol Ideal Gas Constant • If 8.314 is the kPa constant, how much is it for atm? • Convert 8.314 as if it were kPa to atm: • 8.314 / 101.3 = 0.0821 – sound familiar? • Multiply 0.0821 by 760 to get the torr constant: • 0.0821 * 760 = 62.38 – sound familiar? • You have a choice: 1. Memorize only the kPa constant and convert P. 2. Memorize all three and not convert. 3. Memorize only the kPa constant and convert R. Ideal Gas Law Practice • 1 mol of hydrogen gas at 273 K exerts a pressure of 101.3 kPa. How much space does it take up? • PV = nRT • 101.3 V = 1 8.314 273 • V = 22.4 L – Well doesn’t that make sense now? – This shows that a mol of any gas takes up 22.4 L just like we learned, but only at STP. Ideal Gas Law Practice • A 47.3 L container containing 1.62 mol of He is heated until the pressure reaches 1.85 atm. What is the temperature? • R = 8.314 when pressure is kPa, so convert. • 1.85 atm = 187.405 kPa • PV = nRT • 187.405 47.3 = 1.62 8.314 T • T = 658.14 K Ideal Gas Law Practice • Gas Laws Packet – Ideal Gas Law page, odd numbers Dalton’s Law of Partial Pressures • Proposed by John Dalton, 1803: • For a mixture of gases in a container, the total pressure equals the sum of the individual gases’ pressures (“partial pressures”). – Useful for calculating pressure of gases collected over water. • Important: For each, P = (nRT)/V: Ptotal P1 P2 P3 ... Dalton’s Law Practice • A mixture of gases at total pressure 120 kPa contains N2, CO2, and O2. The partial pressure of the nitrogen is 43 kPa and the partial pressure of the CO2 is 34 kPa. What is the partial pressure of the O2? • Ptotal = P1 + P2 + P3 • 120 kPa = 43 kPa + 34 kPa + Poxygen • Poxygen = 43 kPa Dalton’s Law Continued • In the same container, however, R, T, and V are all the same, so the only thing that can change is the number of moles (n). • Thus: Ptotal n1RT n 2RT n 3RT = ... V V V Dalton’s Law Practice Deux • A 5 L container is heated to 300 K. Inside are 3 mol of N2, 5 mol of H2, and 1 mol of O2. What is the total pressure? • Start by solving for P for each gas (P = nRT/V): • Pnitrogen = (3 mol)(8.314)(300 K) / (5 L) = 1496.52 kPa • Phydrogen = (5 mol)(8.314)(300 K) / (5 L) = 2494.2 kPa • Poxygen = (1 mol)(8.314)(300 K) / (5 L) = 498.84 kPa • Now add: • 1496.52 + 2494.2 + 498.84 = 4489.56 kPa Dalton’s Law Practice Deux Alternative Solution • A 5 L container is heated to 300 K. Inside are 3 mol of N2, 5 mol of H2, and 1 mol of O2. What is the total pressure? • Add up the moles (5 + 1 + 3 = 9) and do one equation: • Ptotal = (9 mol)(8.314)(300 K) / (5 L) = 4489.56 kPa The Mole Fraction • Out of Dalton’s Law, chemists pulled other relationships. • One of them is the mole fraction – the ratio of moles of the substance to total moles. – It’s exactly like a percentage, except not multiplied by 100. It’s a proportion. – The symbol used is the Greek letter chi - c n1 P1 c = n total Ptotal Mole Fraction Practice • Part 1: The partial pressure of nitrogen in air is 592 torr. Air pressure is 752 torr, what is the mole fraction of nitrogen? • c = P1/Ptotal • c = 592/752 • c = 0.787 • This is like saying nitrogen is 78.7% of the air, which it is. Mole Fraction Practice • Part 2: What is the partial pressure of nitrogen if the container holding the air is compressed to 5.25 atm? (c was 0.787; P was 752 torr) • Nitrogen’s mole ratio (determined using pressure) was 0.787. • 5.25 * 0.787 = 4.13 atm • It doesn’t matter how the pressure changes; nitrogen is still about 0.787 “percent” of the total. Mole Fraction Practice • Gas Laws Extension Packet – #1 Gas Density • Let’s start with a story: • It’s May 25th, 2014. My wife and I are playing cards with a group of people at our friend Erin’s house. • Erin has a Great Dane named Karma, a sweet old lady of 10 years. – She also weighs more than me. The dog’s a horse. • Karma is sleeping near our card game. • Karma farts when she sleeps. Gas Density • As you might imagine, they are SBDs. We can’t see or hear them; we can only smell them. • As Karma lets one rip, you can see the movement of the fart across the circle of people. – First, people nearest ground zero hold their noses, then those further away, and finally those on the other side of the room get watery eyes. • This is the diffusion of a gas. • As you might imagine, it depends on the density or mass of the gas being passed. Gas Density • There are two ways to think of gas density. – One uses a new, optional formula. The other uses PV=nRT. Let’s start with the latter. • PV=nRT – Suppose you hold everything constant but make temperature go up. What happens to volume? • Volume has to go up too. – What happens to density when volume goes up but mass is unchanged? • Density goes down. Get it? Gas Density • The other way to think of this is through an equation. • This is a derivation of the ideal gas law*, but the short story is: • M = molar mass (NOT mass) • D = density DRT M P *Derivation on next slide. Gas Density Formula Derivation You don’t need to know this. • If Density = Mass / Volume… • …and Molar Mass = Mass / Number of Moles… This is the important – Let M stand for molar mass… one, yo. – …and m is mass, and n = PV / RT…then… m M = (PV/RT) Ptotal mRT m RT DRT M = PV V P P (n1 + n 2 + n 3 ...)RT = V Ptotal (n total)RT = V Gas Density • Other ways to think of gas density: – Warm air rises. • Warmer air (increase in temp) has a lower density than cooler air. – Carbon monoxide leaks tend to affect those sleeping upstairs. • CO is less dense than normal air and rises. – Radon leaks tend to stay in basements. • Rn is a big, heavy gas. • Or just remember: – Temperature increases? Density decreases. – Pressure increases? Density increases. Gas Density Practice Above the level of test questions. • What is the density of ammonia (NH3) at 23 °C and 735 torr? • 23 °C = 296 K • R = 8.314 when pressure is kPa so convert: • 735 torr = 97.97 kPa • M = (D R T) / P • • • • 17.031 = (D*8.314*296) / 97.97 17.031 = D*2460.944 / 97.97 17.031 = D*25.119 D = 0.678 g/L Gas Density Practice • What is the density (g/L) of ammonia (NH3) at 23 °C and 735 torr in a 2 L container? • 23 °C = 296 K • R = 8.314 when pressure is kPa so convert: • 735 torr = 97.97 kPa • PV = nRT • (97.97)(2) = (n)(8.314)(296) • n = 0.0796 mol • 0.0796 mol NH3 = 1.36 g NH3 • D = g/L • D = 1.36 / 2 • D = 0.68 g/L Gas Density Challenger Above the level of test questions. • A compound has the empirical formula CHCl. A 256 mL flask at 100 °C and 750 torr contains 0.80 g of the gaseous compound. What is the molecular formula? • • • • • • • • CHCl molar mass = 48.4689 g/mol R = 8.314 when pressure is torr so convert: 750 torr = 99.967 kPa M = (mRT) / (PV) M = (0.80*8.314*373) / (99.967*0.256) Molar Mass = 96.942 96.942/48.4689 ≈ 2 Molecular Formula: C2H2Cl2 Back to farting… • Diffusion, describes the spreading out and mixing of gases. • Effusion is the movement of a gas through a small hole in its container. • The rate of diffusion/effusion increases with temperature. • Small molecules diffuse/effuse faster than large molecules. Effusion • Think of effusion like elementary school kids… • …when an ice cream truck pulls up outside the school at the end of the day. – They’ll frantically try to squeeze through the doorway to get outside. • If you did the same thing, it would take you longer. You’re bigger, so you can’t effuse as quickly. – However, temperature increases effusion rate, so you could always light a fire in the room. That would help. Graham’s Law of Effusion • Proposed by Thomas Graham, 1840s: • The rate of effusion of a gas is inversely proportional to the square root of the gas’s molar mass (because light gases go faster). • M1 = Molar mass of gas 1; V1 = Speed of gas 1 – Hint: Make M1 and V1 for the lighter/faster gas. • M2 = Molar mass of gas 2; V2 = Speed of gas 2 V1 M2 = V2 M1 Graham’s Law Practice 1 • Which gas moves faster: Hydrogen or chlorine, and by what factor? – Molar mass of Hydrogen: 1.0079 g/mol – Molar mass of Chlorine: 35.45 g/mol V1 M2 = V2 M1 70.90Cl VH = VCl 2.02 H • √35.10 = 5.92, so H travels 5.92 times faster than Cl. Thank goodness. – Notice something else: The problem only compared rates of effusion; it didn’t provide actual numbers. What about V1/V2? • We actually don’t need to do anything with it. • 5.92 is equal to V1/V2. • We don’t know how fast each gas is going. We just know hydrogen is going faster. Graham’s Law Practice 2 • Calculate the ratio of the velocity of helium atoms to fluorine molecules at the same temperature. – Molar mass of He: 4.0026 g/mol – Molar mass of F2: 37.996 g/mol V1 M2 = V2 M1 VHe 37.996 F = VF 4.0026 He • √9.49 = 3.08, so He travels about 3 times faster than F. – The ratio is 3.08 : 1. Graham’s Law Practice 3 • Helium travels 5.73 times faster than an unknown monatomic gas. What is the unknown gas? – Molar mass of He: 4.0026 g/mol V1 M2 = V2 M1 M2 5.73 = 4.0026 He M2 5.73 = 4.0026 He 2 • M2 = 131.42 g/mol, so the unknown gas is Xe. Graham’s Law Hints 1. As mentioned before, make Gas 1 the lighter/faster one. 2. Don’t think of V1/V2 as something to solve. Solving the right side gives a ratio represented as a single number. That number represents how much faster gas 1 is than gas 2. 3. You’ll normally find a rate >1, which means Gas 1 is faster than Gas 2. A rate <1 means Gas 1 is slower than Gas 2; =1 means they’re equal. Gas Diffusion Video • CrashCourse – Passing Gasses – Effusion, Diffusion, and the Velocity of a Gas Test Question Review Expect something similar on the test. • We’ll review this in our “molar volume of a gas” lab, but for now, here’s some good practice: – Many gases can be collected over water; that is, the gas is produced in a reaction under water and the bubbles are collected in the space above the surface at the top. – Unfortunately, that means that some of the water also evaporates, creating two combined pressures: 1. 2. Pressure of the water vapor (the vapor pressure). Pressure of the desired gas. – You’ll need to adjust some numbers a bit to remove water vapor pressure. • KEY: The atmospheric pressure equals the two pressures listed above. https://lh3.googleusercontent.com/0fEKN5qdk9NRwAg8qLCq1cfXdlZttYeClID0oBpvvvhtKKiFrwg57xNyBzNrJUH7CJZWilmMUVOjaNA4VgE3IVZuriBBlcEpRaP6jPXcXb8YxjzVUM Test Question Review Expect something similar on the test. • Imagine atmospheric pressure (total pressure) is 102 kPa and the vapor pressure of water is 21 kPa. The temperature is 300 K. A reaction is performed using 0.5 mol Li: – 2Li + 2HCl 2LiCl + H2 • How much hydrogen (in liters) could be collected? • Many students first think that you can just solve the stoichiometry problem and convert to liters. • The problem is that a mole of gas only takes up 22.4 L at standard temperature and pressure, which we don’t have here. 2Li + 2HCl 2LiCl + H2 • Solve first for mol of H2 created (stoichiometry). • 0.5 mol Li = 0.25 mol H2 • Now let’s eliminate pressure due to H2O. • Patm = Pwater + Phydrogen • 102 kPa = 21 kPa + Phydrogen • Phydrogen = 81 kPa • Now the standard PV = nRT • • • • • P = 81 kPa V=? n = 0.25 mol R = 8.314 LkPa / Kmol T = 300 K • Solving for V gets you 7.70 L. Solving These Problems • At some point, these problems are almost always solved by doing these things: – Determine moles of product gas expected to form. • Stoichiometry/mole ratio. – Eliminate effect of water vapor pressure. • Dalton’s Law: Subtract water vapor pressure from atmospheric pressure. – Solve for new volume formed. • Ideal Gas Law or Combined Gas Law. Gas Collection Practice • Gas Laws Extension Packet – #5, Part 3, A-D (on Page 3) – The skeleton equation is KClO3 KCl + O2 Mystery Gas Law Practice • A sample of gas has a volume of 4.18 L at 29 °C and 732 torr. What would its volume be at 24.8 °C and 756 torr? – Gas Law of Choice: Combined • Problem has no mention of moles and a lot of before/after. • 29 °C = 302 K; 24.8 °C = 297.8 K • P1V1 / T1 = P2V2 / T2 • (732 * 4.18) / 302 = (756 * V2) / 297.8 • V2 = 3.99 L Mystery Gas Law Practice • Gas Laws Packet – Assorted page (all) – Identify the gas law that you use! • STP Reminder: • 1 atm • 0 °C One last bit of reinforcement… • To really make sure we get this stuff, let’s use a gizmo on ExploreLearning. – It’s been a while, huh? • What you’ll do is each get a computer, log in to Quia AND ExploreLearning, then use Quia to guide you through a gizmo on ExploreLearning. – Answering the questions as you go. • Quia quiz is called “Boyle’s Law and Charles’ Law Gizmo.” • [Log-in Instructions] Closure • For the balloon-in-a-flask demo, what was the most applicable law? – A: Boyle’s Law – B: Charles’ Law – C: Gay-Lussac’s Law – D: Combined Gas Law – E: Ideal Gas Law – F: Dalton’s Law Closure • For the balloon-in-a-flask demo, what was the most applicable law? – A: Boyle’s Law – B: Charles’ Law • Because we were dealing with changing V and T. – C: Gay-Lussac’s Law • Because we were dealing with changing T and the effects of P. – D: Combined Gas Law – E: Ideal Gas Law – F: Dalton’s Law Concepts • Gas Laws Extension Packet – Relationships section Concepts • How would the boiling point of water at high altitudes (like Vail, Colorado) compare with the boiling point of water at sea level? Explain why. – High altitude boiling points are lower because liquids boil when vapor pressure equals atmospheric pressure. Less pressure from the air on the liquid means less energy is needed for liquid molecules to escape to gases. Concepts • [see Space Jump video] • Why will a person’s blood boil at extremely high altitudes such as those experienced by Felix Baumgartner during his record-breaking skydive (if he did not have a pressurized suit)? – With lower atmospheric pressure, there is less force keeping molecules in the liquid state. As a result, they will start to become gas, despite the temperature being extremely low. Concepts • [see Space Jump video] • Why did the balloon get bigger as it rose? – Boyle’s Law or Charles’ Law: With lower atmospheric pressure, the molecules in the balloon could push further out, expanding the volume. Concepts • A pressure cooker is a sealed metal pot that can cook foods under high pressures. Would your food cook faster or slower compared to using a regular pot? Explain why. – Faster – higher pressure raises boiling points, meaning the food can be cooked at a higher temperature and thus finish more quickly. – Additionally, added pressure increases temperature on its own (Gay-Lussac’s Law). Concepts • If the volume of a container of gas is reduced, what will happen to the pressure inside the container? – Boyle’s Law – the pressure will increase. Same amount of gas but a smaller space to occupy means increased collisions. Concepts • Why does the pressure inside a container of gas increase if more gas is added to the container? Assume a rigid container. – More molecules means more collisions between molecules.