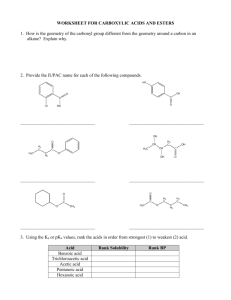
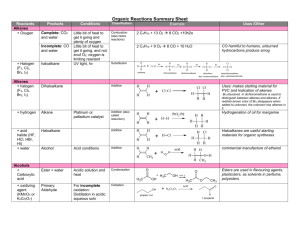
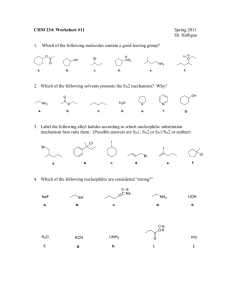
Organic Reactions
advertisement

Organic Reactions A detailed study of the following: Dehydration Synthesis Addition Free Radical Reactions Substitution (SN1 & SN2) Elimination (E1 & E2) Dehydration Synthesis • A reaction involving the formation of a single product through the formation & removal of water. • These reactions usually involve reactions between an alcohol and something else. What can be made using this process? • • • • Alcohol + alcohol Ether* Alcohol + acid Ester* Alcohol + ammonia Amine Alcohol + Acid Amide • * These are discussed further Dehydration of Alcohols to form Ethers • Simple, symmetrical ethers can be formed from the intermolecular acid-catalyzed dehydration of 1° (or methyl) alcohols (a “substitution reaction”) • 2° and 3° alcohols can’t be used because they eliminate (intramolecular dehydration) to form alkenes OH H3O+ + OH + O heat H2O •Unsymmetrical ethers can’t be made this way because a mixture of products results: + OH + CH3-OH H3O heat + O + O O Mechanism of Formation of Ethers from Alcohols • First, an alcohol is protonated by H3O+ • Next, H2O is displaced by another alcohol (substitution) • Finally, a proton is removed by H2O to form the product H OH H O + + O H H O H H H H OH O O + O + H H H H H O H + H H O O + O H H Combustion of alkanes • Alkanes are unreactive as a family because of the strong C–C and C–H bonds as well as them being nonpolar compounds. At room temperature alkanes do not react with acids, bases, or strong oxidizing agents. • Alkanes do undergo combustion in air (making them good fuels): 2C2H6(g) + 7O2(g) 4CO2(g) + 6H2O(l) H = –2855 kJ • Complete combustion produced carbon dioxide and water while incomplete may produces a combination of carbon monoxide, carbon and water in addition to carbon dioxide. Carbon dioxide contributes to global warming while carbon monoxide is toxic; hemoglobin binds to carbon monoxide in preference to oxygen causing suffocation and even death. Products of combustion Complete combustion produces: carbon dioxide water vapour while incomplete may produces a combination of : carbon monoxide carbon water vapour carbon dioxide. Carbon dioxide contributes to global warming. Carbon monoxide is toxic; hemoglobin binds to carbon monoxide in preference to oxygen causing suffocation and even death. Alkane Substitution Reaction • In the presence of light alkanes undergo substitution reaction with halogens. RH + Br2 RBr + HBr • In a substitution reaction, one atom of a molecule is removed and replaced or substituted by another atom or group of atoms. • Mechanism of subtitution reaction involves free radicals. Free Radical Substitution reaction UV CH3CH 2CH 2CH 2CH 2CH3 Br2 CH3CH 2CH 2CH 2CH 2CH 2 Br HBr 1-bromohexane For a reaction between an alkane and bromine to occur, C-H and Br-Br bonds must break. The C-H bond is stronger than Br-Br bond Therefore, the reaction proceeds by first the breakage of Br-Br bond, which is brought about by UV light. Br-Br bond can be broken in one of two ways. Br2 2Br . UV or UV Br2 Br : Br Free Radical Substitution reaction When the bond is broken, either •the bond pair can be equally shared between the two atoms producing two bromine atoms (called free radicals), or •The bond pair goes with one atom producing a positive and a negatively charged ions of bromine. The first type of bond breakage producing free radicals is referred to as a homolytic fission and the second heterolytic fission. •Homolytic fission because the bond pairs are equally distributed, or particles that are the same in every way is produced. •homolytic fission of the halogen takes place. In the next step, the free radical removes a hydrogen atom from the alkane forming hydrogen bromine and a free radical of the alkane. CH3CH2CH2CH2CH2CH2-H + Br• CH3CH2CH2CH2CH2CH2• + HBr Free Radical Substitution reaction • The free radical goes on to react with a molecule of chlorine and regenerate another chlorine free radical. CH3CH2CH2CH2CH2CH2• + Br2 CH3CH2CH2CH2CH2CH2Br + Br• And so on. Because this reaction, once initiated, can keep itself going is referred to as a chain reaction. The reaction can conducted with any halogen and the mechanism would be the same. Not only that, more than one hydrogen can be substituted. UV CH3CH 2CH 2CH 2CH 2CH3 2Br2 CH3CH 2CH 2CH 2CH 2CHBr2 2HBr 1,1 dibromohexane Mechanism of chlorination of methane CHAIN REACTION 1. Initiation .. : Cl .. .. Cl : .. light 2 .. : Cl . .. a free radical “dissociation” R E P E A T I N G S T E P S 2. Chain Propagation (first step) + CH3 H .. : Cl . .. H .. Cl : .. + . CH3 methyl radical “hydrogen abstraction” 3. Chain Propagation (second step) . CH3 + .. : Cl .. .. Cl : .. CH3 .. Cl : .. + .. : Cl . .. feeds back into step two Mechanism of chlorination of methane 4. Termination Steps .. 2 : Cl . .. CH3. .. : Cl . .. + + . CH 3 . CH 3 “recombinations” .. : Cl .. .. Cl : .. CH3CH3 .. : Cl CH3 .. These steps stop the chain reaction Reactions of Alkenes: Addition Reactions Hydrogenation of Alkenes – addition of H-H (H2) to the π-bond of alkenes to afford an alkane. The reaction must be catalyzed by metals such as Pd, Pt, Rh, and Ni. H H H C + C Pd/C H H EtOH H H C-C π-bond = 243 KJ/mol H-H = 435 KJ/mol H C C H H H H°hydrogenation = -136 KJ/mol H C-H = 2 x -410 KJ/mol = -142 KJ/mol • The catalysts is not soluble in the reaction media, thus this process is referred to as a heterogenous catalysis. • The catalyst assists in breaking the -bond of the alkene and the H-H -bond. • The reaction takes places on the surface of the catalyst. Thus, the rate of the reaction is proportional to the surface area of the catalyst. 14 14 • Carbon-carbon -bond of alkenes and alkynes can be reduced to the corresponding saturated C-C bond. Other -bond bond such as C=O (carbonyl) and CN are not easily reduced by catalytic hydrogenation. The C=C bonds of aryl rings are not easily reduced. O O H2, PtO2 ethanol O C5H11 OH H2, Pd/C CH3(CH2)16CO2H Linoleic Acid (unsaturated fatty acid) Steric Acid (saturated fatty acid) O O OCH3 H2, Pd/C OCH3 ethanol C H2, Pd/C N C N ethanol 15 15 Heats of Hydrogenation -an be used to measure relative stability of isomeric alkenes H H3C H CH3 cis-2-butene H°combustion : -2710 KJ/mol H H3C H H2, Pd CH3 cis-2-butene H H3C CH3 H trans-2-butene trans isomer is ~3 KJ/mol more stable than the cis isomer -2707 KJ/mol H2, Pd H CH3 CH3CH2CH2CH3 H3C H trans-2-butene H°hydrogenation: -119 KJ/mol -115 KJ/mo trans isomer is ~4 KJ/mol more stable than the cis isomer The greater release of heat, the less stable the reactant. 16 16 Heats of Hydrogenation of Some Alkenes Alkene H2C=CH2 H H H3C H monosubstituted H H° (KJ/mol) 136 125 - 126 H 117 - 119 H3C CH3 H CH3 disubstituted H3C H3C H 114 - 115 H 116 - 117 H3C H H3C H H3C CH3 H3C CH3 H3C CH3 trisubstituted tetrasubstituted 112 110 17 17 Electrophilic Addition of Hydrogen Halides to Alkenes C-C -bond: H°= 368 KJ/mol C-C -bond: H°= 243 KJ/mol -bond of an alkene can act as a nucleophile!! Electrophilic addition reaction H H Br C C H + H-Br H nucleophile H H C C H H H electrophile Bonds broken C=C -bond 243 KJ/mol H–Br 366 KJ/mol Bonds formed H3C-H2C–H -410 KJ/mol H3C-H2C–Br -283 KJ/mol calc. H° = -84 KJ/mol expt. H°= -84 KJ/mol 18 18 Reactivity of HX correlates with acidity: HF << HCl < HBr < HI fastest Regioselectivity of Hydrogen Halide Addition: H Markovnikov's Rule Br H H-Br C H slowest R R R C H R C R C C H C R R C C H H H H H-Br H H-Br Br H R C C H R H Br H R C C R R H + + + H Br R C C H H H none of this H Br R C C H R H none of this H Br R C C R R H none of this H R C C R' H H-Br Br H R C C R H H + H Br R C C R' H H Both products observed For the electrophilic addition of HX across a C=C bond, the H (of HX) will add to the carbon of the double bond with the most H’s (the least substitutent carbon) and the X will add to the carbon of 19 19 the double bond that has the most alkyl groups. Mechanism of electrophilic addition of HX to alkenes Regioselectivity determined by Markovnikov’s rule – which can be explained by comparing the stability of the intermediate carbocations 20 20 For the electrophilic addition of HX to an unsymmetrically substituted alkene: • The more highly substituted carbocation intermediate is formed. • More highly substituted carbocations are more stable than less substituted carbocations. (hyperconjugation) • The more highly substituted carbocation is formed faster than the less substituted carbocation. Once formed, the more highly substituted carbocation goes on to the final product more rapidly as well. 21 21 Carbocation Rearrangements in Hydrogen Halide Addition to Alkenes - In reactions involving carbocation intermediates, the carbocation may sometimes rearrange if a more stable carbocation can be formed by the rearrangement. These involve hydride and methyl shifts. H C H3C C H3C Cl H H-Cl C H C H3C H3C H H H H C C H H + H ~ 50% expected product H C H3C C H3C Cl CH3 H C H H-Cl H3C C C Cl H H3C H C C CH3 H H H H ~ 50% H C H3C C H3C H3C H + C H3C H3C H H C C Cl H H Note that the shifting atom or group moves with its electron pair. A MORE STABLE CARBOCATION IS FORMED. 22 22 Free-radical Addition of HBr to Alkenes H3CH2C H3CH2C R R R H H C H C H C R C R C R C C H C H H H-Br Br H H3CH2C C C H H H H H-Br Br H H3CH2C C C H H H peroxides (RO-OR) H-Br C H C H C R C R' H ROOR (peroxides) H H-Br ROOR H H-Br ROOR H H-Br ROOR + + H Br H3CH2C C C H H H none of this H Br H3CH2C C C H H H Polar mechanism (Markovnikov addition) Radical mechanism (Anti-Markovnikov addition) none of this Br H R C C H H H none of this Br H R C C H R H none of this Br H R C C R R H none of this Br H R C C R H H + + H Br R C C H H H H Br R C C H R H + H Br R C C R R H + H Br R C C R' H H Both products observed The regiochemistry of HBr addition is reversed in the presence of peroxides. Peroxides are radical initiators - change in mechanism 23 23 The regiochemistry of free radical addition of H-Br to alkenes reflects the stability of the radical intermediate. H H R C• R C• H Primary (1°) R R C• R < Secondary (2°) R < Tertiary (3°) 24 Acid-Catalyzed Hydration of Alkenes The addition of water (H-OH) across the -bond of an alkene to give an alcohol; opposite of dehydration H3C C H3C CH2 H2SO4, H2O H3C H3C H3C C OH This addition reaction follows Markovnikov’s rule The more highly substituted alcohol is the product and is derived from The most stable carbocation intermediate. Reactions works best for the preparation of 3° alcohols 25 p. 91a Mechanism for this reaction is the reverse of the acid-catalyzed dehydration of alcohols: 27 6.11: Thermodynamics of Addition-Elimination Equlibria H3C H2SO4 C CH2 H3C C H3C H3C + H2O H3C Bonds broken C=C -bond 243 KJ/mol H–OH 497 KJ/mol OH Bonds formed H3C-H2C–H -410 KJ/mol (H3C)3C–OH -380 KJ/mol calc. H° = -50 KJ/mol G° = -5.4 KJ/mol H° = -52.7 KJ/mol S° = -0.16 KJ/mol How is the position of the equilibrium controlled? Le Chatelier’s Principle - an equilibrium will adjusts to any stress The hydration-dehydration equilibria is pushed toward hydration (alcohol) by adding water and toward alkene (dehydration) by removing water. 28 The acid catalyzed hydration is not a good or general method for the hydration of an alkene. Oxymercuration: a general (2-step) method for the Markovnokov hydration of alkenes H H C C4H9 H 1) Hg(OAc)2, H2O C H Hg(OAc) C H H O C H3C C C4H9 H Ac= acetate = OH O 2) NaBH4 OH C C4H9 H C H H NaBH4 reduces the C-Hg bond to a C-H bond 29 Addition of Halogens to Alkenes X2 = Cl2 and Br2 X2 X X (vicinal dihalide) C C C C alkene 1,2-dihalide Stereochemistry of Halogen Addition - 1,2-dibromide has the anti stereochemistry Br Br + + Br2 Br Br not observed CH3 Br Br2 H CH3 Br 30 Substitution Reaction with Halides (1) bromomethane If concentration of (1) is doubled, the rate of the reaction is doubled. If concentration of (2) is doubled, the rate of the reaction is doubled. (2) methanol If concentration of (1) and (2) is doubled, the rate of the reaction quadruples. Substitution Reaction with Halides (1) (2) bromomethane methanol Rate law: rate = k [bromoethane][OH-] this reaction is an example of a SN2 reaction. S stands for substitution N stands for nucleophilic 2 stands for bimolecular Mechanism of SN2 Reactions Alkyl halide The rate of reaction depends on the concentrations of both reactants. When the hydrogens of bromomethane are replaced with methyl groups the reaction rate slow down. The reaction of an alkyl halide in which the halogen is bonded to an asymetric center leads to the formation of only one stereoisomer Relative rate 1200 40 1 ≈0 Mechanism of SN2 Reactions Hughes and Ingold proposed the following mechanism: Transition state Increasing the concentration of either of the reactant makes their collision more probable. Mechanism of SN2 Reactions Steric effect Energy activation energy: G2 activation energy: G1 reaction coordinate reaction coordinate Inversion of configuration (R)-2-bromobutane (S)-2-butanol Factor Affecting SN2 Reactions The leaving group - HO HO HO HO relative rates of reaction + RCH2I RCH2OH + I + RCH2Br RCH2OH + Br + RCH2Cl RCH2OH + Cl + RCH2F RCH2OH + F pKa HX 30 000 10 000 200 1 -10 -9 -7 3.2 The nucleophile In general, for halogen substitution the strongest the base the better the nucleophile. pKa Nuclephilicity SN2 Reactions With Alkyl Halides an alcohol a thiol an ether a thioether an amine an alkyne a nitrile Substitution Reactions With Halides 1-bromo-1,1-dimethylethane 1,1-dimethylethanol Rate law: If concentration of (1) is doubled, the rate of the reaction is doubled. If concentration of (2) is doubled, the rate of the reaction is not doubled. rate = k [1-bromo-1,1-dimethylethane] this reaction is an example of a SN1 reaction. S stands for substitution N stands for nucleophilic 1 stands for unimolecular Mechanism of SN1 Reactions Alkyl halide Relative rate The rate of reaction depends on the concentrations of the alkyl halide only. ≈0* When the methyl groups of 1-bromo1,1-dimethylethane are replaced with hydrogens the reaction rate slow down. ≈0* The reaction of an alkyl halide in which the halogen is bonded to an asymetric center leads to the formation of two stereoisomers 12 1 200 000 * a small rate is actually observed as a result of a SN2 Mechanism of SN1 Reactions nucleophile attacks the carbocation slow C-Br bond breaks fast Proton dissociation Mechanism of SN1 Reactions Rate determining step G Carbocation intermediate R++ X+ R-OH2 R-OH Mechanism of SN1 Reactions Inverted configuration relative the alkyl halide Same configuration as the alkyl halide Factor Affecting SN1 reaction Two factors affect the rate of a SN1 reaction: • The ease with which the leaving group dissociate from the carbon • The stability of the carbocation The more the substituted the carbocation is, the more stable it is and therefore the easier it is to form. As in the case of SN2, the weaker base is the leaving group, the less tightly it is bonded to the carbon and the easier it is to break the bond The reactivity of the nucleophile has no effect on the rate of a SN1 reaction Comparison SN1 – SN2 SN1 SN2 A two-step mechanism A one-step mechanism A unimolecular rate-determining step A bimolecular rate-determining step Products have both retained and inverted configuration relative to the reactant Product has inverted configuration relative to the reactant Reactivity order: 3o > 2o > 1o > methyl Reactivity order: methyl > 1o > 2o > 3o Elimination Reactions 1-bromo-1,1-dimethylethane 2-methylpropene Rate law: rate = k [1-bromo-1,1-dimethylethane][OH-] this reaction is an example of a E2 reaction. E stands for elimination 2 stands for bimolecular The E2 Reaction A proton is removed Br- is eliminated The mechanism shows that an E2 reaction is a one-step reaction Elimination Reactions 1-bromo-1,1-dimethylethane 2-methylpropene Rate law: If concentration of (1) is doubled, the rate of the reaction is doubled. rate = k [1-bromo-1,1-dimethylethane] If concentration of (2) is doubled, the rate of the reaction is not doubled. this reaction is an example of a E1 reaction. E stands for elimination 1 stands for unimolecular The E1 Reaction The base removes a proton The alkyl halide dissociate, forming a carbocation The mechanism shows that an E1 reaction is a two-step reaction Products of Elimination Reaction 30% 2-bromobutane 50% 80% 2-butene 20% 1-butene The most stable alkene is the major product of the reaction for both E1 and E2 reaction For both E1 and E2 reactions, tertiary alkyl halides are the most reactive and primary alkyl halides are the least reactive The greater the number of alkyl substituent the more stable is the alkene Competition Between SN2/E2 and SN1/E1 SN1 SN2 E1 E2 rate = k1[alkyl halide] + k2[alkyl halide][nucleo.] + k3[alkyl halide] + k2[alkyl halide][base] • SN2 and E2 are favoured by a high concentration of a good nucleophile/strong base • SN1 and E1 are favoured by a poor nucleophile/weak base, because a poor nucleophile/weak base disfavours SN2 and E2 reactions Competition Between Substitution and Elimination • SN2/E2 conditions: In a SN2 reaction: 1o > 2o > 3o In a E2 reaction: 3o > 2o > 1o 10% 90% 75% 25% 100% Competition Between Substitution and Elimination • SN1/E1 conditions: All alkyl halides that react under SN1/E1 conditions will give both substitution and elimination products (≈50%/50%) Summary of Elimination & Substitution Reactions • Alkyl halides undergo two kinds of nucleophilic subtitutions: SN1 and SN2, and two kinds of elimination: E1 and E2. • SN2 and E2 are bimolecular one-step reactions • SN1 and E1 are unimolecular two step reactions • SN1 lead to a mixture of stereoisomers • SN2 inverts the configuration od an asymmetric carbon • The major product of a elimination is the most stable alkene • SN2 are E2 are favoured by strong nucleophile/strong base • SN2 reactions are favoured by primary alkyl halides • E2 reactions are favoured by tertiary alkyl halides