polyatomic compounds
advertisement

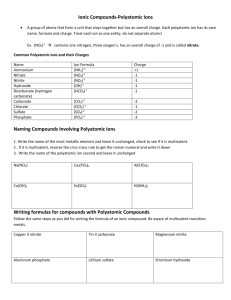
Starter #28 What is the symbol and name for the compound formed from mixing Cobalt (+2) and chlorine POLYATOMIC COMPOUNDS (Challenge) Chapter 5.9 Polyatomic Compounds Are groups of atoms that tend to stay together and carry an overall ionic charge. Example: Nitrate ion When a compound containing this ion is dissolved in water, the positive metal ion and the nitrate ion separate from each other, but the nitrate ion itself stays together as a unit surrounded by water molecules. Some ionic compounds are made of polyatomic ions which is an ion with more than one element in it. Looking at the top center of your Periodic Chart of Ions handout you will see examples of these Examples: CO32- Carbonate NO31- Nitrate Note: you will know the difference between a regular non-metal ion and a polyatomic ion by the fact that there is no “ide” ending on the polyatomic ion. WRITING FORMULAS FOR POLYATOMIC COMPOUNDS All the compound formula calculations and compound naming procedures are the same as for regular ionic compounds. EXAMPLE: What is the formula for the ionic compound formed by sodium and sulfate ion? STEPS 1) Write the symbols of the metal and of the polyatomic group Na SO4 2) Write the ionic charges Na+1 SO4-2 3) Choose the number of ions to balance the charge Na+1 Na+1 SO4-2 STEPS 4) Write the formula using subscripts. Na2SO4 You could use the criss-cross rule if you wanted to. EXAMPLE 2 What is the formula for lead (IV) carbonate? 1) Pb CO3 2) Pb+4 CO3-2 3) criss-cross rule 4) Pb2(CO3)4 (must reduce) 5) Pb(CO3)2 Starter #29 What is the name and formula for the compound formed by calcium and nitrate? Try This PAGE 198; 3 abce, 4 abd Questions 3. Write the formula for: a) sodium phosphate b) calcium sulfate c) potassium chlorate e) beryllium nitrate 4. Name the following compounds: a) K2C03 b) Na2SO4 d) AgNO3 SOLUTIONS 3a) Na3PO4 3b) CaSO4 3c) KClO3 3e) Be(N03)2 4a) Potassium Carbonate 4b) Sodium Sulfate 4d) Silver Nitrate