Ben Hwang Wiki Assignment

Ben Hwang
Tuesday March 04, 2014
Intramolecular and Intermolecular
Forces and Molecular Polarity
Agenda:
Homework:
Read pages 96 -98, 109 -117 on Intramolecular and Intermolecular Hydrogen Bonding,
Effect on the properties of Ionic, true Covalent and Polar Covalent Compounds
Finish ALL worksheets, and answer questions on page 73 # 7 and 8, Page 113 (Mini
Investigation), Page 115 # 4 and 5, Page 118 #1-3, Page 89 – 92 #36 – 38, 40, 65 (On inter/intra), 69, and 70
Go over tutorial 1and table 3 on page 106, page 228 (Grade 12 textbook)
Electronegativity and physical Properties:
Helps explain the properties of Compounds (ex. in Labs)
For instance, in HCl, there are partial charges that keep the molecule together
NaCl has similar properties to those of HCl, but in NaCl the attraction is greater. Δ EN for NaCl is 2.9 vs. 0.9 for
HCl
In general, since Ionic compounds tend to have high electro negativities, Ionic compounds tend to have high melting points
Example: For each, pick one that that would have the lower boiling point: a.)
CaCl
2
, CaF
2
b.) KCl,
LiBr
c.) H
2
0,
H
2
S
For all, the lower the Δ EN of the two molecules is the one which will have the lower boiling point. This is not always true as there are other factors that can be involved. (Ex.
Atomic size). Useful to compare atoms/molecules of the same size
Polar Molecules:
Polar bonds MAY cause entire molecule to be polar
A polar molecule is a molecule where there is unequal distribution of electrons. This results in one end of the molecule to become positive, and the other end to become negative
Non Polar molecules are molecules where the electron distribution is shared equally, resulting in no localized charges
Bond dipoles can/may not cancel out producing molecules that are either polar if they cancel, or non-polar if they do not cancel out
Example:
BF
3 has three polar bonds but BF
3 is not a polar molecule. The reason for this is because the molecule is symmetrically distributed, so the polar bonds actually cancel out making it a non-polar molecule.
H
2
O is held together with O-H bonds which are polar. The molecule is unequal and the molecule is set up asymmetrically creating dipole forces. The molecule is set up with a positive side on one end, and a negative side on the other making H
2
O or water very polar.
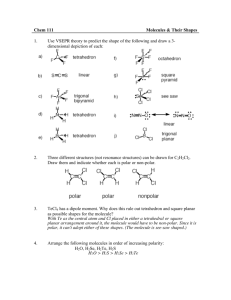
Predicting Molecular Polarity – General Steps:
Step 1: Draw a reasonable Lewis diagram
Step 2: Identify bonds either polar or non-polar ( Δ EN lower or equal to 0.4 is non-polar)
Step 3: Examine central atom if there is one and examine electrons around it. If there are no lone pairs and bonds are the same it is non – polar. If there are polar bonds, and molecule is not symmetrical, it is probably polar
Step 4: Draw a geometric sketch of molecule
Step 5: Determine symmetry. Use arrows to show polar bonds pointing to more electronegative element (Use different lengths of arrows to show relative polarities of the bonds). If arrows are symmetrical and arrows are same length, molecule is non-polar. If arrows are different lengths and do not balance, or arrangement is asymmetrical, molecule is polar.
Example:
Decide whether the following molecules are polar or non-polar by drawing Lewis Dot Diagrams: a.) CO
2
b.) HCN
CO
2 has two polar bonds, yet the entire molecule is not polar. The reason for this is because the molecular structure of the molecule is symmetrical which cancels out the polar bonds
HCN has a polar bond, between C and N, and has a non-polar bond between H and C. There is one lone pair of electrons on the outside of
N, and the side of H is positive. This molecule is therefore a polar molecule.
Helpful Links:
http://www.chemguide.co.uk/atoms/bonding/electroneg.html
http://www.youtube.com/watch?v=PcJRH2ojkZA