Experiment 21
advertisement

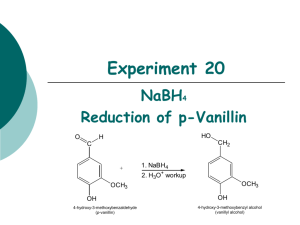
Experiment 20 NaBH4 Reduction of p-Vanillin O HO H CH2 C + 1. NaBH4 2. H3O+ workup OCH3 OCH3 OH 4-hydroxy-3-methoxybenzaldehyde (p-vanillin) OH 4-hydroxy-3-methoxybenzyl alcohol (vanillyl alcohol) Objectives: To synthesize vanillyl alcohol by reduction using NaBH4. To isolate and purify the product by vacuum filtration. To analyze the purity of products using TLC, HPLC and melting point analysis. To characterize the reactants and products using 1H-NMR and IR spectroscopy. Before coming to lab… Review these techniques: Vacuum filtration TLC Analysis HPLC analysis Melting point analysis CHEMICAL EQUATION This is provided as a solution. Be sure you understand how to calculate theoretical yield! O HO H CH2 C + 1. NaBH4 2. H3O+ workup OCH3 OCH3 OH 4-hydroxy-3-methoxybenzaldehyde (p-vanillin) OH 4-hydroxy-3-methoxybenzyl alcohol (vanillyl alcohol) MECHANISM O H C O H H B H H C HO H CH2 H O H H H + H2O + B(OH)3 (from 6M HCl) OH 4-hydroxy-3-methoxybenzaldehyde (p-vanillin) OCH3 OCH3 OCH3 OH OH 4-hydroxy-3-methoxybenzyl alcohol (vanillyl alcohol) REDUCING AGENTS Reducing agents cause a reaction resulting in a product containing more bonds from carbon to hydrogen (or fewer bonds to oxygen). NaBH4 (sodium borohydride) is a versatile and useful reducing agent in organic chemistry, however will only reduce carbonyl groups of aldehydes and ketones. LAH (lithium aluminum hydride) is another common reducing agent, however use of this reducing agent will result in the reduction of many other carbonyl containing compounds, including esters, carboxylic acids H H Li+ Na+ and amides. H B H H Sodium borohydride H Al H H Lithium aluminum hydride H Hydride ion TLC STAIN—2,4-DNP R H C HN NH 2 HN O NO2 N NO2 + H R aldehyde or ketone NO2 2,4-DNP (red-orange) NO2 product hydrazone (yellow-orange) 2,4-DNP is a TLC stain used to detect aldehydes and ketones OVERVIEW Dissolve p-vanillin in ethanol. Add NaBH4 solution drop-wise via addition funnel while stirring. Add 6M HCl to hydrolyze intermediate, forming alcohol product. Isolate solid product by vacuum filtration. Prepare TLC and HPLC samples—submit HPLC sample for analysis. Perform TLC experiment on product, staining with 2,4-DNP stain. Secure product and allow to dry until next lab period. OVERVIEW AT THE BEGINNING OF THE NEXT LAB PERIOD… Obtain final mass of product and calculate % yield. Perform melting point analysis. Analyze HPLC results. SYNTHESIS Mix p-vanillin and ethanol in a water cooled 25 mL flask with stir bar until dissolved. Set up apparatus shown. Add NaBH4/NaOH solution to separatory funnel. Add a few ice cubes to the water bath. Begin adding NaBH4/NaOH solution SLOWLY. 7 8 6 5 4 3 2 9 1 0 1 7 8 9 6 5 11 4 3 2 1 SYNTHESIS Remove water bath. Stir 5 min. Return ice water bath. Add 6M HCl until pH = 6. Stir for 10 min. in ice bath. PRODUCT ISOLATION & PURIFICATION Suction filter to isolate solid. Rinse solid in funnel with icecold water. Dry solid under vacuum for a few minutes. PRODUCT ANALYSIS Prepare TLC and HPLC sample of solid product. Secure remaining product and submit to instructor to dry until next lab period. filter paper Submit HPLC sample for analysis. Prepare TLC plate using sample and provided standards. Develop plate and visualize under UV. Stain plate with 2,4-DNP TLC stain. A B C PRODUCT ANALYSIS—TLC TLC Analysis Used to identify and determine purity of products at the end of the experiment. 2,4-DNP will be used as a stain to detect the presence/absence of the aldehyde functional group. Table 20.1 Rf values should always be recorded to 2 decimal places! Never more, never less! Compound p-vanillin Vanillyl Alcohol Standard Rf Sample Rf TLC Diagram Sketch the plate as much to scale as possible! Include cm measurements for all spots and solvent front! PRODUCT ANALYSIS—IR IR Analysis IR spectra of reactants and products can be used to determine the presence and absence of certain types of functional groups which indicate the conversion of one compound to another during the course of the synthesis. Table 20.2 Functional Group OH stretch C-O stretch C=O stretch Base Values p-vanillin Vanillyl alcohol Frequency (cm-1) Frequency (cm-1) Frequency (cm-1) 3200-3600 1000-1300 1680-1740 Notice that the product should have 2 entries for O-H and C-O! PRODUCT ANALYSIS—NMR NMR Analysis 1H-NMR spectra of reactants and products can be used to determine the presence and absence of certain types of signals which indicate the conversion of one compound to another during the course of the synthesis. O This is where the major change is! H1 C 6 2 HO 7 CH2 1 6 2 5 5 OCH3 OCH3 3 3 OH 4 OH 4 Tables 20.4 and 20.5 Table 20.4 Theoretical yield (g) Actual yield (g) Percent yield Product Appearance Experimental Melting Point (oC) Table 20.5 Atom Economy (%) Experimental Atom Economy (%) “Eproduct” Cost per synthesis ($) Cost per gram ($/g) PRODUCT ANALYSIS—HPLC HPLC Analysis Used at the end of the experiment to identify and quantify compounds present during the synthesis, as well as the purity of the final products from each step. Table 20.6 Compound p-vanillin Vanillyl alcohol HPLC Retention Times (min) Standard Sample PRODUCT ANALYSIS—HPLC HPLC Analysis Used at the end of the experiment to identify and quantify compounds present during the synthesis, as well as the purity of the final products from each step. Compound p-vanillin Vanillyl alcohol HPLC Retention Times (min) Standard Sample SAFETY CONCERNS Ethanol, ethyl acetate, hexane, and acetone are all FLAMMABLE materials. Sodium borohydride and 2,4-DNP are TOXIC in large concentrations. Sulfuric acid, used to prepare 2,4-DNP stain, is CORROSIVE. WASTE MANAGEMENT o LIQUID WASTE : Place all liquid waste into this container including filtrates and aqueous washes from extraction. o GLASS WASTE: Place used TLC and melting point capillary tubes in this container. o PAPER WASTE: Place any gloves, TLC plates, filter papers, paper towels, etc. in the yellow trashcan. CLEANING Any glassware used to contain only volatile organic solvents can simply be rinsed with wash acetone. All other glassware should be cleaned with soap, water and brush, then rinsed with wash acetone or hand dried. LABORATORY NOTEBOOK (Pre-lab) • OBJECTIVE (Must clearly state…) • What compounds will be made and how • What will be investigated with the compounds • CHEMICAL EQUATION • Include the general chemical equation from the top of page 169 from hondout. • TABLE OF PHYSICAL DATA (Complete the following table using MSDS sheets from http://hazard.com/msds or www.chemexper.com ONLY. Wikipedia is unacceptable.) Compound p-vanillin Sodium borohydride ethanol Sodium hydroxide Vanillyl alcohol Hydrochloric acid Ethyl acetate Hexane 2,4-dinitrophenylhydrazine MW (g/mol) bp(oC) mp (oC) X X X X X X X X X X X X X X X X X X X • REFERENCE TO PROCEDURE d (g/mL) X X X X X X (Must include…) • full title including edition and author names • numbers where actual procedure can be found OSHA HAZARDS LABORATORY NOTEBOOK (In-lab) • DATA/CALCULATIONS Initial mass of p-vanillin used Volume of NaBH4 solution used Volumes of ethanol and 6M HCl used Weights of small filter paper, filter + product, and final product mass HPLC vial slot # TLC diagram, including all cm measurements Example Rf calculation Theoretical yield calculation (not just value!) % yield calculation (not just value!) Atom economy calculation (not just value!) Experimental atom economy calculation (not just value!) “Eproduct” calculation (not just value!) Cost per synthesis calculation (not just value!) Cost per gram calculation (not just value!) Cost per synthesis calculation Cost per gram calculation EXPERIMENTAL PROCEDURE • In paragraph form, describe the procedure that you actually followed during the lab. • Paragraph must be written in PAST TENSE, PASSIVE VOICE. • Include any volumes or weights of chemicals used during the experiment. • Include any mistakes, accidents or observations if applicable.