chap13b - WordPress.com
advertisement

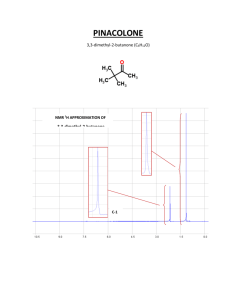
NUCLEAR- Study of nuclear spins MAGNETIC- Under the influence of applied magnetic field RESONANCE- Record the resulting resonance in nuclear spin through the absorption of RF INTEGRATION NMR Spectrum of Phenylacetone O CH2 C CH3 RECALL from last time Each different type of proton comes at a different place . You can tell how many different types of hydrogen there are in the molecule. INTEGRATION OF A PEAK Not only does each different type of hydrogen give a distinct peak in the NMR spectrum, but we can also tell the relative numbers of each type of hydrogen by a process called integration. Integration = determination of the area under a peak The area under a peak is proportional to the number of hydrogens that generate the peak. Benzyl Acetate The integral line rises an amount proportional to the number of H in each peak METHOD 1 integral line integral line 55 : 22 : 33 = 5:2:3 simplest ratio of the heights Benzyl Acetate (FT-NMR) Actually : 5 58.117 / 11.3 = 5.14 2 21.215 / 11.3 = 1.90 3 33.929 / 11.3 = 3.00 O CH2 O C CH3 METHOD 2 digital integration assume CH3 33.929 / 3 = 11.3 Integrals are good to about 10% accuracy. Modern instruments report the integral as a number. DIAMAGNETIC ANISOTROPY SHIELDING BY VALENCE ELECTRONS Diamagnetic Anisotropy The applied field induces circulation of the valence electrons - this generates a magnetic field that opposes the applied field. valence electrons shield the nucleus from the full effect of the applied field magnetic field lines Bo applied B induced (opposes Bo) fields subtract at nucleus Diamagnetic Anisotropy The applied field induces circulation of the valence electrons - this generates a magnetic field that opposes the applied field. valence electrons shield the nucleus from the full effect of the applied field magnetic field lines Bo applied B induced (opposes Bo) fields subtract at nucleus PROTONS DIFFER IN THEIR SHIELDING All different types of protons in a molecule have a different amounts of shielding. They all respond differently to the applied magnetic field and appear at different places in the spectrum. This is why an NMR spectrum contains useful information (different types of protons appear in predictable places). DOWNFIELD Less shielded protons appear here. SPECTRUM UPFIELD Highly shielded protons appear here. It takes a higher field to cause resonance. CHEMICAL SHIFT PEAKS ARE MEASURED RELATIVE TO TMS Rather than measure the exact resonance position of a peak, we measure how far downfield it is shifted from TMS. reference compound tetramethylsilane “TMS” CH3 CH3 Si CH3 CH3 Highly shielded protons appear way upfield. TMS shift in Hz downfield n 0 Chemists originally thought no other compound would come at a higher field than TMS. REMEMBER FROM OUR EARLIER DISCUSSION field strength frequency hν = γ B o 2π constants ν = ( K) Bo Stronger magnetic fields (Bo) cause the instrument to operate at higher frequencies (ν). NMR Field Strength 1.41 T 2.35 T 7.05 T 1H Operating Frequency 60 Mhz 100 MHz 300 MHz HIGHER FREQUENCIES GIVE LARGER SHIFTS The shift observed for a given proton in Hz also depends on the frequency of the instrument used. Higher frequencies = larger shifts in Hz. TMS shift in Hz downfield n 0 THE CHEMICAL SHIFT The shifts from TMS in Hz are bigger in higher field instruments (300 MHz, 500 MHz) than they are in the lower field instruments (100 MHz, 60 MHz). We can adjust the shift to a field-independent value, the “chemical shift” in the following way: parts per million chemical = shift δ shift in Hz = spectrometer frequency in MHz = ppm This division gives a number independent of the instrument used. A particular proton in a given molecule will always come at the same chemical shift (constant value). HERZ EQUIVALENCE OF 1 PPM What does a ppm represent? 1H Operating Frequency 60 Mhz 100 MHz 300 MHz 7 6 1 part per million of n MHz is n Hz Hz Equivalent of 1 ppm n MHz ( 60 Hz 100 Hz 300 Hz 5 4 3 2 1 1 = n Hz ) 6 10 0 ppm Each ppm unit represents either a 1 ppm change in Bo (magnetic field strength, Tesla) or a 1 ppm change in the precessional frequency (MHz). NMR Correlation Chart -OH -NH DOWNFIELD DESHIELDED UPFIELD SHIELDED CHCl3 , H TMS 12 11 10 9 8 7 6 H RCOOH RCHO C=C 5 4 CH2F CH2Cl CH2Br CH2I CH2O CH2NO2 3 2 1 0 d (ppm) CH2Ar C-CH-C CH2NR2 C CH2S C-CH2-C C C-H C=C-CH2 C-CH3 CH2-CO Ranges can be defined for different general types of protons. This chart is general, the next slide is more definite. APPROXIMATE CHEMICAL SHIFT RANGES (ppm) FOR SELECTED TYPES OF PROTONS R-CH3 R-CH2-R R3CH 0.7 - 1.3 1.2 - 1.4 1.4 - 1.7 R-C=C-C-H O 1.6 - 2.6 R-C-C-H O 2.1 - 2.4 RO-C-C-H O 2.1 - 2.5 HO-C-C-H 2.1 - 2.5 N C-C-H 2.1 - 3.0 R-C C-C-H 2.1 - 3.0 C-H R-C C-H 2.3 - 2.7 1.7 - 2.7 R-N-C-H 2.2 - 2.9 R-S-C-H 2.0 - 3.0 I-C-H 2.0 - 4.0 Br-C-H 2.7 - 4.1 Cl-C-H 3.1 - 4.1 RO-C-H 3.2 - 3.8 HO-C-H O 3.2 - 3.8 R-C-O-C-H 3.5 - 4.8 O2N-C-H 4.1 - 4.3 F-C-H 4.2 - 4.8 R-C=C-H 4.5 - 6.5 H 6.5 - 8.0 O R-C-N-H 5.0 - 9.0 O R-C-H 9.0 - 10.0 O R-C-O-H 11.0 - 12.0 R-N-H 0.5 - 4.0 Ar-N-H 3.0 - 5.0 R-S-H R-O-H 0.5 - 5.0 Ar-O-H 4.0 - 7.0 1.0 - 4.0 YOU DO NOT NEED TO MEMORIZE THE PREVIOUS CHART IT IS USUALLY SUFFICIENT TO KNOW WHAT TYPES OF HYDROGENS COME IN SELECTED AREAS OF THE NMR CHART C-H where C is CH on C attached to an aliphatic acid aldehyde benzene alkene next to C-H COOH CHO CH =C-H electronega- pi bonds tive atom X=C-C-H X-C-H 12 10 9 7 6 4 3 2 0 MOST SPECTRA CAN BE INTERPRETED WITH A KNOWLEDGE OF WHAT IS SHOWN HERE DESHIELDING AND ANISOTROPY Three major factors account for the resonance positions (on the ppm scale) of most protons. 1. Deshielding by electronegative elements. 2. Anisotropic fields usually due to pi-bonded electrons in the molecule. 3. Deshielding due to hydrogen bonding. We will discuss these factors in the sections that follow. DESHIELDING BY ELECTRONEGATIVE ELEMENTS DESHIELDING BY AN ELECTRONEGATIVE ELEMENT d- Cl d+ C d- electronegative element H d+ Chlorine “deshields” the proton, that is, it takes valence electron density away from carbon, which in turn takes more density from hydrogen deshielding the proton. NMR CHART “deshielded“ protons appear at low field highly shielded protons appear at high field deshielding moves proton resonance to lower field Electronegativity Dependence of Chemical Shift Dependence of the Chemical Shift of CH3X on the Element X Compound CH3X Element X Electronegativity of X Chemical shift d most deshielded CH3F CH3OH CH3Cl CH3Br CH3I CH4 (CH3)4Si F O Cl Br I H Si 4.0 3.5 3.1 2.8 2.5 2.1 1.8 4.26 3.40 3.05 2.68 2.16 0.23 0 TMS deshielding increases with the electronegativity of atom X Substitution Effects on Chemical Shift most deshielded most deshielded CHCl3 CH2Cl2 CH3Cl 7.27 5.30 3.05 ppm -CH2-Br 3.30 -CH2-CH2Br 1.69 The effect increases with greater numbers of electronegative atoms. -CH2-CH2CH2Br 1.25 ppm The effect decreases with incresing distance. ANISOTROPIC FIELDS DUE TO THE PRESENCE OF PI BONDS The presence of a nearby pi bond or pi system greatly affects the chemical shift. Benzene rings have the greatest effect. Ring Current in Benzene Circulating electrons H Bo H Deshielded fields add together Secondary magnetic field generated by circulating electrons deshields aromatic protons ANISOTROPIC FIELD IN AN ALKENE protons are deshielded Deshielded fields add H shifted downfield C=C H Bo H H secondary magnetic (anisotropic) field lines ANISOTROPIC FIELD FOR AN ALKYNE H C C H Bo Shielded fields subtract hydrogens are shielded secondary magnetic (anisotropic) field HYDROGEN BONDING HYDROGEN BONDING DESHIELDS PROTONS R O H H O H O R The chemical shift depends on how much hydrogen bonding is taking place. Alcohols vary in chemical shift from 0.5 ppm (free OH) to about 5.0 ppm (lots of H bonding). R Hydrogen bonding lengthens the O-H bond and reduces the valence electron density around the proton - it is deshielded and shifted downfield in the NMR spectrum. SOME MORE EXTREME EXAMPLES O H O C R R C O H O Carboxylic acids have strong hydrogen bonding - they form dimers. With carboxylic acids the O-H absorptions are found between 10 and 12 ppm very far downfield. H3C O O H O In methyl salicylate, which has strong internal hydrogen bonding, the NMR absortion for O-H is at about 14 ppm, way, way downfield. Notice that a 6-membered ring is formed.