STUDY GUIDE FOR FINAL Thursday March 29th, 2012 You may
advertisement

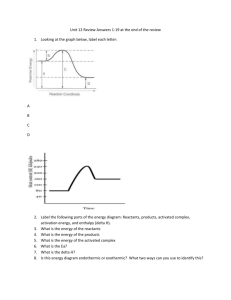
STUDY GUIDE FOR FINAL Thursday March 29th, 2012 You may use a note card; all equations will be given Thermochemistry: Chapter 17 online text. Sections: 17.1, 17.2, 17.3, 17.4 (Please read if you are confused and need more help) Also if you are struggling on any problems PLEASE come in for help. Vocabulary to Know: Not all the answers are here, look them up yourself Heat Temperature Specific Heat Calorie Heat of fusion Heat of vaporization Endothermic Exothermic Enthalpy Heat of reaction Hess’s Law Thermochemistry calculations/problems: 1.) Heat equation: q=mC∆T Be able to solve for heat (q) or mass, or specific heat (C), or ∆T Example: How much heat energy is needed to raise the temperature of a 55 g sample of aluminum from 22.4⁰C to 94.6 ⁰C? The specific heat of aluminum is .89 J/g⁰C? Answer: 3534.19 J 2.) Heat of fusion/heat of vaporization: Q= m∆Hfus or Q=m∆Hvap Be able to solve for heat (q) Example: How many kJ of heat are absorbed when .46 g of chloroethane (C2H5Cl), vaporizes. The molar heat of vaporization of chloroethane is 26.4 kJ/mol. Answer: .188 kJ Additional problem(s): Chapter 17; # 71 (Answer 23.6 kJ), pg. 539 #1 (Answer a) 3.) Calorimetry problems: (Two step calculation) Q=mC∆T for water and C= Q/m∆T for the metal Example: A 30.5 g piece of metal at 100⁰C is placed in 80 ml of water at 24⁰C in an insulated calorimeter. The specific heat of water is 1.0 cal/g⁰C. The metal and water came to equilibrium at a final temperature of 30⁰C. What is the specific heat of the metal? Answer: First find the heat gained by the water q=mC∆T, then find the specific heat of the metal C=q/m∆T, .26cal/g⁰C. 4.) Heating curve: Be able to label the states of matter, where the temperature is changing and where a phase change is occurring. Refer to worksheet. What happens to the temperature during a phase change? Answer: temperature remains constant 5.) Calculating the heat of the reaction using heats of formation: Remember elements in their natural states, like the gases O2, F2, H2 do not take any energy to form so therefore are 0 kJ/mol. Example: What is the heat of the reaction ∆Hrxn for the following reaction if the heat of formation of O2 is 0 kJ/mol, the heat of formation of CO is -110.5 kJ/mol, and the heat of formation of CO2 is -393.5 kJ/mol. 2CO + O2 → 2CO2 Answer: Remember ∆Hrxn= ∆Hf(Products)- ∆Hf(Reactants) = -566 kJ Additional problems: Chapter 17, # 60 (answer 0), 69 a) -221 kJ, b) -196 kJ, c) -904.6 kJ in Online text 6.) Be able to solve for amount of heat produced using Stoichimetry and thermo chemical equations: Example: When carbon disulfide is formed from its elements, heat is absorbed. Calculate the amount of heat (in kJ) is absorbed when 5.66 grams of carbon disulfide is formed. C(s) + 2S(s) → CS2 (l) ∆Hrxn = 89.3 kJ Answer: 6.63 kJ Additional problem(s): Chapter 17; #19 (Answer 372 kJ) in online text 7.) Hess’s Law: Be able to calculate the heat of the reaction from heat of the reaction from intermediate steps. Example: Calculate the enthalpy change (∆Hrxn) in kJ for the following reaction. 2Al(s) + Fe2O3 (s) → 2Fe(s) + Al2O3(s) The intermediate steps are: 2Al(s) + 3/2 O2(g) → Al2O3(s) ∆Hrxn = -1669.8 kJ 2Fe (s) + 3/2 O2(g) → Fe2O3(s) ∆Hrxn = -824.2 kJ Answer: -845.6 kJ Additional Problem(s): Chapter 17; #59 (Answer 30.2 kJ), 66 (Answer -750 kJ), 74 (Answer 180 kJ), 81 (Answer -1207 kJ), 84 (Answer -137 kJ) in Online text Rates of Reactions: Chapter 18 online, Sections 18.1, 18.5 Vocabulary Not all the answers are here, look them up yourself Collision theory Activated complex-transition state Activation Energy (Ea) Catalyst Inhibitor Rate Law Rate of reactions: 1.) Be able to interpret potential energy diagrams for Endothermic and exothermic Reactions. What are the differences between endothermic/exothermic diagrams. Refer to homework sheet and notes. Answer: Endothermic: Reactants are lower in energy than products, so need to absorb energy. Exothermic: Reactants are higher in energy that products so need to release energy. 2 .) Know the four factors affecting the rate of the reactions. -Temperature -Surface Area/Particle Size - Concentration - Catalyst 3.) Know how a catalyst effects a reaction. A catalyst speeds up a reaction by lowering the Activation Energy. A catalyst remains unchanged in a reaction. 4.) Rate Law: Be able to determine the rate order and constant using experimental data. Rate: k[A]x [B]y [C]z Remember k is the rate constant and the x, y, z are the rate order of that particular reactant you are trying to determine. Example: Determine the rate order of the individual reactants and the overall rate of this reaction using the following data: A + B→C+ D Rate: k [A]x [B]y Experiment 1 2 3 Answer: Initial rate (M/s) .0050 .080 .0050 Initial [A] .10 .40 .10 Initial [B] .10 .10 .20 Rate= .5[A]2[B]0 Note: doubling (2?)the concentration quadruples(=4) the initial rate of formation of product so it is 2nd order (exponent) dependant on chemical A; when you change the concentration of Chemical B it doesn’t affect the rate at all so it is “powerless” (and anything to the zero poer is “one” so you can skip even writing it)