Exam 1 Review
advertisement

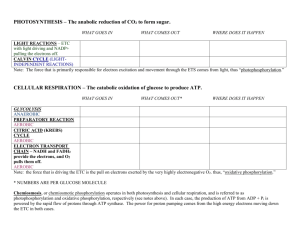
Exam 1 Review Ch. 1- Science of Biology Kingdoms of life include Animalia, Plantae, Protista, Fungi, Bacteria & Archaea What is homeostasis? Why is it essential to all living things? What are the underlying themes of biology Evolution by cooperation; In many cases, organisms have changed (evolved) in response to one another. Distinguish between hypothesis and theory Deductive vs Inductive reasoning; Scientists use inductive reasoning when performing scientific processes. Review the proper order of the Scientific Process Review the properties shared among living organisms CFCs (Chlorofluorocarbons) act as a catalyst to destroy the ozone which increases UV passage to Earth Definition of Heredity Definition of Theory Symbiosis and the origin of eukaryotic cells; Be able to describe the process by which eukaryotes evolved from prokaryotes Ch. 3- Chemistry of Life Be able to distinguish between the atomic number and atomic mass. What do these numbers indicate? Subatomic Particles o Protons- found in the nucleus; positively charged (+1) o Neutrons- found in the nucleus; no charge (0) o Electrons- found in orbitals; negatively charged (-1) Ions- atoms with a positive or a negative charge o Cations- positively charged ion o Anions- negatively charged ion Orbitals pH (acids vs bases); What is the pH scale. Recall that acids have a pH that is less than 7 (<7) and bases have a pH that is greater than 7 (>7). Ionic bonds have a tendency to break when dissolved in water. Ionic bonds form when two atoms are bonded together due to a transfer of electrons. Covalent bonds are formed when to atoms are bonded together due to the sharing of electrons. Review electrons and how they distinguish the properties of atoms o If you change the number of electrons of an atom, the properties of the atom change!! 1 electrons with a filled outer energy level are stable; electrons without a filled outer energy level are reactive. o 2 electrons fill the first energy level o 8 electrons fill the second and third energy level Ch. 4- Molecules of Life The four essential macromolecules are o Carbohydrates- composed of monosaccharides. When many monosaccharides join together a polysaccharide is formed. o Lipids- include fats, phospholipids, and steroids; composed of a glycerol and fatty acids o Proteins- built out of amino acids. Amino acids are joined together by a peptide bond to build a polypeptide chain. o Nucleic Acids- build out of nucleotides; examples of nucleic acid include DNA and RNA Dehydration synthesis is the process by which organic macromolecules such as polysaccharides are made by joining individual monosaccharides. Dehydration synthesis involves the removal of water. Basic structure of proteins Levels of protein structure o Primary- sequence of amino acids o Secondary- formation of alpha helix or beta-pleated sheets o Tertiary- three-dimensional shape of a protein o Quaternary- combining of two polypeptide chains DNA vs RNA o DNA- double stranded molecule containing the sugar deoxyribose. DNA has the four bases Adendine, Thymine, Guanine, and Cytosine o RNA- single stranded molecule containing the sugar ribose. RNA has the four bases Adendine, Uracil, Guanine, and Cytosine Properties of phospholipids- composed of a polar region with two nonpolar fatty acid tails Ch. 5- Cells Cell size (surface area to volume ratio); Why are cell small? Nuclear Envelope; What is its function? Where is it found? Active Transport (low to high concentration); Are molecules moving with the concentration gradient or against it? Prokaryotic cells vs Eukaryotic- Prokaryotes appeared on Earth first and are believed to have given rise to Eukaryotes. Prokaryotic cells are the simplest cells. Hypotonic vs Hypertonic o A hypertonic solution such as salt water will cause a cell to shrivel because there is net movement of water out of the cell. o A hypotonic solution such as pure water will cause a cell to swell and possibly burst because there is a net movement of water into the cell. Cell theory o All organisms are composed of cells 2 o Cells are the basic unit of structure and function (smallest living things) o All cells come from pre-existing cells Know the function of ribosomes in protein synthesis. Carbohydrates and lipids are manufactured in the smooth Endoplasmic reticulum Why are chloroplasts and mitochondria believed to have been free-living bacterial cells? The golgi body transports proteins out of the cell. Endocytosis vs exocytosis Active transport vs passive transport o Active transport involves a transmembrane protein and energy to pump molecules from low to high concentration. o Passive transport may occur through the membrane or membrane proteins and does not require energy because molecules are moving from high to low concentration. Chemiosmosis Bacterial cells do not have membrane bound organelles. Osmosis is the diffusion of water Diffusion Transmembrane channel proteins help polar molecule and ions move into and out of the cell. Different kinds of microscopes o Compound light microscope o Electron microscope o Phase-Contrast microscope What is involved in exocytosis? Ch. 6-Energy and Life Oxidations vs Reduction o Oxidation- loss of an electron o Reduction- Gain of an electron Enzymes are used to speed up the rate of a reaction; required to make and break molecules in biological organisms. Enzymes lower the activation energy of reactions. Entropy is the measure of disorder in a system. Most of the cell’s energy is stored in ATP 1st law of thermodynamics- “Energy cannot be created or destroyed” Ch. 7- Photosynthesis: Acquiring Energy from the Sun Review the equation for photosynthesis CAM photosynthesis is apparent in areas with extreme heat like the desert. Photosynthesis takes place in the chloroplasts Where within the chloroplast do the light-independent reactions take place? Electromagnetic- full range of energy in sunlight What is the function of Calvin Cycle? 3 Chemiosmosis- process by which ATP is generated when protons move through ATP Synthase. Where are ATP and NADPH molecules generated? What gas is taken from the air during photosynthesis to make glucose? Ch. 8- How Cells Harvest Energy from Food Stages of Cellular Respiration o Glycolysis o Krebs Cycle o Electron Transport Chain The electron carriers in the Krebs Cycle are NADH and FADH2; Electron carriers deliver their electrons to the electron transport chain. Oxygen is the final Electron Acceptor in aerobic respiration Which part of cellular respiration produces the largest amount of ATP? What is the purpose of Fermentation Function of ETC Respiration vs Photosynthesis 4