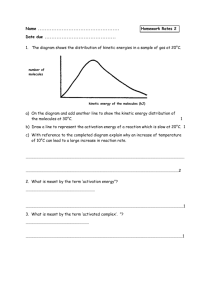
Kinetics and Equilibrium review answers
1) 1
2) 3
3) 1
4) 4
5) 2
6) 2
7) 2
8) 3
9) 1
10) 4
11) 3
12) 2
13) 3
14) 1
15) 3
16) 2
17) 2
18) 1
19) 2
20) 4
21) 2
22) 1
23) Entropy increases due to the particles having more freedom of movement
24) Solid sodium chloride has no mobile ions
25) Heat flows from the water to the salt
26) D
27) C
28) KNO3 any dissolving process that is endothermic
29) C2H2
30) 3
31) Arrow 2 gets shorter
32) A moles of nitrogen increase
B moles of hydrogen decrease
C no effect a catalyst causes an increase in rate not a shift
33) endothermic the products have more energy than reactants
34)
35) Decreasing the concentration of carbon dioxide causes a shift right as carbonic acid
decomposes to form carbon dioxide and water
36) +4
37) a catalyst lowers activation energy
38) 1, 3, 2, 3
39)
40) 1808 – 273 = 1535
 0
0