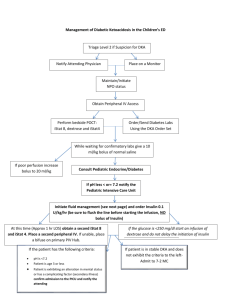
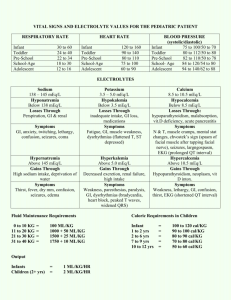
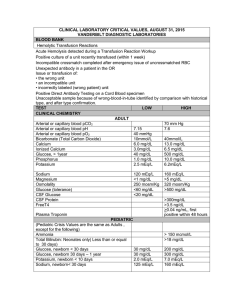
CHEM 115 QUIZ #1 1/18/00
advertisement

CHEM 115 QUIZ #5 Answer Key 1. 2/29/00 A solution contains 40.0 mEq/L of Cl- and 15.0 mEq/L of HPO42-. If Na+ is the only cation in the solution, what is the Na+ concentration in mEq/L? Any solution must be overall neutral, i.e., the total concentration of anions must equal the total concentration of cations, concentrations expressed in mEq/L which takes account of the different charges on the ions. Total concentration of anions = 40.0 mEq/L + 15.0 mEq/L = 55.0 mEq/L This must equal the total concentration of cations which is Na+, so Na+ concentration = 55.0 mEq/L 2. How much KCl is needed to prepare 50.0 mL of a 5.00% (m/v) KCl solution? mass KCl = 50.0 mL solution x (5.00 g KCl/100 mL solution) = 2.5 g KCl Note how the concentration is used as a conversion factor that allows us to convert volume (in mL) to mass (in grams). 3. At 30oC, the solubility of CuSO4 is 25.0 g/100 g of water. How many grams of CuSO 4 are needed to make a saturated CuSO4 solution with 500.0 g of water at 30oC? Mass of CuSO4 = 500.0 g water x (25.0 g CuSO4/100 g water) = 125 g CuSO4 Note how the solubility is used as a conversion factor that allows us to convert mass of water (solvent) to mass of solute (CuSO4).