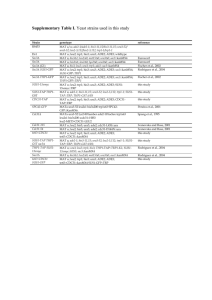
Classical genetics
advertisement

Classical genetics • Monofactorial cross, ie. A (wt) x a (mut) – 2:2 pattern of segregation – if 4:0 -> mitochondrial • Complementation / non-compl. (in Dipl.) movie Tetrad dissection Micromanipulator for Tetrad Dissection Movie Tetrad analysis movie Spore/Row A B C D Tetrad No 1 2 3 4 5 6 Genetic Linkage Bifactorial Cross Possibilities: AB x ab LEU2 ura3∆ x leu2∆ URA3 1) A and B are on the same chromosome - no crossover - one crossover - double crossover 2) A and B are on different chromosomes - no crossover between marker and cen - one crossover between marker and cen - two crossover between marker and cen Meiosis -> 4 spores • Yeast tetrad analysis is nothing else than just watching the outcome of meiosis • Diploid 2n, two sets of chromosomes • DNA is replicated resulting in two chromosomes with two identical chromatids each • The chromosomes align and can undergo recombination • The first meiotic division will separate the chromosomes from each other • The second meiotic division will separate the chromatids, ie. each spore represents essentially one chromatid The outcome of a cross 1a) LEU2 and URA3 are close together on the same chromosome LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 • If there is no crossover occurs between the two markers all haploid spores will look like the parental haploid strains • There are only two different types of spores, i.e. (leuplus ura-minus) and (leu-minus ura-plus) spores • Hence such a tetrad is called a parental ditype PD Crossover 1b) LEU2 and URA3 are close together on the same chromosome and a crossover occurs between them LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 ura3 LEU2 URA3 leu2 ura3 leu2 URA3 • In this case we will get spores that look like the parental haploids but also spores that have new combinations of the two markers • There are four different types of spores • Hence such a tetrad is called a tetratype T Double crossover 1c) LEU2 and URA3 are close together on the same chromosome and two crossover occur between them such that four DNA strands are involved LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 URA3 LEU2 URA3 leu2 ura3 leu2 ura3 • In this case we will get only spores that look different from the parental haploids • There are two different types of spores • Hence such a tetrad is called non parental ditype NPD The probability of a crossover depends on gene distance LEU2 ura3 LEU2 ura3 leu2 URA3 URA3 • One crossover -> T • Double crossover -> NPD • No crossover -> PD leu2 LEU2 URA3 LEU2 URA3 leu2 ura3 leu2 ura3 • Thus with close linkage: PD > T > NPD and the relative numbers can be used to map genetic distances. For mapping one analyzes hundreds of tetrads from the same cross. This has been done extensively in the past and the last genetic map from 1995 comprised about 1,000 locations cM is a measure for genetic distance between two markers LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 URA3 LEU2 URA3 leu2 ura3 leu2 ura3 To generate new combination of mutations (such as leu2 ura3) one will have to dissect the more tetrads the closer the two genes are, and this can be estimated based on the physical distance (in kb), which relates well to the genetic distance (in cM, centi Morgan). For two close genes (1cM, i.e. 1% recombinant spores) one would have to dissect at least 25 tetrads, statistically Thomas Hunt Morgan • Drosophila geneticist, Nobel price 1933 • Centiomorgan, cM, unit for genetic distance • 1 cM = 1% recombination, 1 in 100 meiotic divisions • Genetic Linkage Map (Alfred Sturtevant) • Yeast 1 cM ca. 3 kb (200-fold less than animal cells, human 1 Mb) 1866-1945, USA Crossing with markers on different chromosomes 2) LEU2 and URA3 are on different chromosomes LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 URA3 LEU2 URA3 leu2 ura3 leu2 ura3 • Different chromosomes assort randomly in the first meiotic division • For this reason two types of tetrads become equally frequent, the parental and the non-parental ditype, PD and NPD • Hence, linked and unlinked genes can easily be distinguished in tetrad analysis because with unlinked genes PD = NPD while with linked genes PD>>NPD. Crossing with markers on different chromosomes 2b) LEU2 and URA3 are on different chromosomes and a crossing over occurs between a centromere and a marker LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 ura3 LEU2 URA3 leu2 ura3 leu2 URA3 • Now the different alleles of URA3 will only be separated in the second meiotic division • The result is a tetratype tetrad T • The above situation means also that if markers are distant from the centromere many Ts will occur while if both markers are close to the centromere few Ts will occur. Crossing with markers on different chromosomes LEU2 ura3 LEU2 ura3 leu2 URA3 leu2 URA3 LEU2 ura3 LEU2 URA3 leu2 ura3 leu2 URA3 • What is the outcome of double cross-overs with four or with three strands? • Due to the possibility of double cross-overs the proportion between different tetrad types for unlinked genes that are not centromere-linked becomes 1:1:4 for PD:NPD:T • This also means that one out of four spores will be recombinant, i.e. in order to obtain the new combination of genes (leu2 ura3) one only needs to dissect one tetrad, statistically Frequency of Tetrades AB x ab If A and B are linked • PD > NPD • The distance of the markers A B determines the number of PD:NPD:T • cM = (100/2)[(T+6NPD)/(PD+NPD+T)] • The number of T is proportional to the map distance Recombination • General recombination = Homologous recombination • Order of events (I meiotic div): – 1) Double strand break – 2) Two chromatids from different chromosomes (maternal/paternal) cross over – 3) Base pairing and strand invasion – 4) Synthesis and gap repair – 5) Resolution of Holiday junction Resolution of Holiday junction Gene conversion Gene conversion Mutagenesis 1. Conventional mutagenesis (MMS, EMS, UV, Transposon) 2. Screen through the knock-out collection or other type of mutant collections For a successful mutagenesis approach one needs a selection or screen to isolate mutants of interest Need to test whether the phenotype of interest is due to a single mutation - tetrad analysis. Recessive Dominant Dominant negative Remember, since yeast can be easily propagated as interconvertible haploids and diploids, studying recessive gene mutations is straightforward. Characterizing mutants 1. Is the mutant phenotype due to a defect in one gene ? => backcross to wt, 2:2 segregation ? If yes, one gene 2. Is the mutation dominant or recessive ? 3. Place the mutants into complementation groups. Usually one complemetation group is equivalent to one gene 4. Identifying/cloning the gene by complementation, or perform genetic linkage analysis to map the gene Dominant and recessive mutations • The dominant or recessive character is revealed by crossing the mutant with the wild type to form a diploid cell • Such diploids are heterozygous, because one chromosome carries the wild type allele and the other one the mutant allele of the gene affected Recessive: wild type phenotype MUT1 MUT1 mut1 • A mutation is dominant when the mutant phenotype is expressed in a heterozygous diploid cell. The diploid has the same phenotype as the haploid mutant • A mutation is recessive when the wild type phenotype is expressed in a heterozygous diploid cell. The diploid has the same phenotype as the wild type mut1 Dominant: mutant phenotype Dominant and recessive mutations • A dominant character can have a number of important reasons, which may reveal properties of the gene product’s function: – The mutations leads to a gain of function, e.g. a regulatory protein functions even without its normal stimulus – The gene product functions as a homo-oligomere and the non-functional monomere causes the entire complex to become non-functional – The gene dosis of one wild type allele is insufficient to confer the wild type phenotype, i.e. there is simply not enough functional gene product (this is rare) Dominant and recessive mutations • The recessive character of a mutation is usually due to loss of function of the gene product • This means that recessive mutations are far more common, because it is simpler to destroy a function than to generate one • Further genetic analysis of the mutant depends on the dominant/recessive character, that is one reason why this step is taken first • In addition, it is useful to do a tetrad analysis of the diploid in order to test that the mutant phenotype is caused by a single mutation, i.e. that the phenotype segregates 2:2 in at least ten tetrads studied; this is important when mutations have been induced by mutagenesis Intragenic complementation • Intragenic complementation is rare, but is does occur • The heterozygous mut1-1/mut1-2 however shows a (partial) wild type phenotype • The occurence of intragenic complementation means that the gene product must be an oligomere • The ”opposite”, non-allelic noncomplementation, can of course also occur: two recessive mutations in two different genes fail to complement. This occurs sometimes when the gene products are involved in the same process or complex and the two functional alleles are just not enough to confer full functionality No functional gene product of MUT1 mut1-1 mut1-1 mut1-2 mut1-2 But a heteromere consisting of Mut1-1p and Mut1-2p can be functional