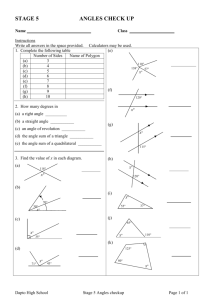
Assignment #12

Chem 1390-099, Fall 2007
Assignment 12
Due Wednesday, 12/5/07, 10 am
Use a separate piece of paper to answer these questions. Please show your work for all calculations and report answers with the correct units and the correct number of significant figures to receive full credit.
Circle your final answers.
1. The phosphorous trihalides (PX
3
, X = halogen) show the following variation in the bond angle X-P-X: PF
3
= 96.3°, PCl
3
= 100.3°, PBr
3
= 101.0°, PI
3
= 102°. The trend is generally attributed to the change in the electronegativity of the halogen.
(a) Assuming that all electron domains (electron pairs) exhibit the same repulsion, what value of the X-P-X angle is predicted by the VSEPR model? Use your textbook.
(b) What is the general trend in the X-P-X angle as the electronegativity increases?
(c) Using the VSEPR model, explain the observed trend in X-P-X angle as the electronegativity of X changes.
(d) Based on your answer to part (c), predict the structure of PBrCl
4
, including estimated bond angles. Give the molecular geometry a name and draw a picture that indicates the three dimensional shape.
2. Tro #9.104, p. 403
3. Tro #9.78, p. 401
4. For each of the following molecules, (i) draw the Lewis dot structure, (ii) determine the 3D molecular geometry, and (iii) determine whether the molecule is polar or nonpolar.
(a) SiCl
4
(b) CF
2
Cl
2
(c) IF
5