Chemistry Worksheet: Foundations & Nomenclature
advertisement

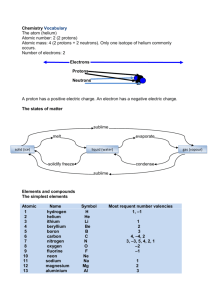
Chapter 1 . Chemical Foundations Q1. How many significant figures are there in each of the following values? a. 6.07 x 10-15 3 b. 0.003840 4 c. 17.00 4 d. 301 3 e. 300. 3 f. 0.00480 3 Q2. Evaluate each of the following and write the answer to the appropriate number of significant figures. a. 212.2 + 26.7 + 402.09 641.0 b. 52.331 + 26.01 – 0.9981 77.34 c. 2.01 x 102 + 3.014 x 103 3215 or 3.215 x 103 Q3. Perform each of the following conversions. a. 294.5 nm to centimeters b. 903.3 angstrom to meters 2.945x10-5 cm 9.033 x 10-8 m Q4. Defination following keywords a. b. c. d. e. f. Macroscopic and Microscopic Law and Theory Precision and Accuracy Homogeneous and Heterogeneous Proton and Neutron Covalent bond and ionic bond Chapter 2 . Naming Simple Compounds Q1. Give the names of the elements that correspond to the following symbols : Sn , Pt, Hg, Mg, K, Mn, Co, Al, P, Na Tin , Platinum , Mercury, Magnesium , Potassium Cobalt , Aluminum , Phosphorus , Sodium Q2. Name each of the following compounds a. Fe2+ Iron(II) or ( Ferrous ion ) b. Fe3+ Iron(III) or ( Ferric ion ) c. S2- Sulfide d. P3- Phosphide a. b. c. d. CsF Cesium Fluoride Ag2S Silver Sulfide TiO2 Titanium Oxide ( dioxide) MnO2 Manganese Oxide a. b. c. d. e. NaHCO3 Sodium hydrogen carbonate NaOCl Sodium hypochlorite HClO Hypochlorous acid H2SO4 Sulfuric acid Na2CO3 Sodium Carbonate Q3. Write the formula for each of the following compounds: a. b. c. d. e. f. g. Silicon dioxide SiO2 Sodium sulfite Na2SO3 Aluminum hydrogen sulfate Al(HSO4)3 Nitrogen trichloride NCl3 Bromous acid HBrO2 Perbromic acid HBrO4 Potassium hydrogen sulfide KHS