Changing the landscape for clinical research in BC.
advertisement

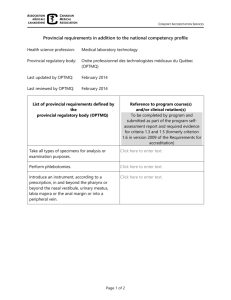
Changing the landscape for clinical research in BC. www.bccrin.ca www.bccrin.ca www.bccrin.ca Research Community www.bccrin.ca www.bccrin.ca www.bccrin.ca www.bccrin.ca www.bccrin.ca Set strategy to achieve a more harmonized approach to research contracts, leading to faster time to study execution. Memorandum of Understanding UBC, BCCA, CFRI, FHA, PHCRI, VCHRI Harmonization of the negotiation and acceptance of industry sponsored trials Industry Forum (May 2011) Key Performance Metrics Identify and track Business of Clinical Trials Increase business acumen Streamlining of costs Study start up mCTA (October 2011 to March 2012) www.bccrin.ca www.bccrin.ca Anonymous, on-line survey 1000 participants across BC Part of a broader strategy to engage British Columbians in the clinical research process Data will be used to improve recruitment and retention outcomes for clinical research sites in BC. Development of strategies designed to address provincial participant perspectives and concerns. www.bccrin.ca Benchmark current levels of investment in clinical research activities within the province, including sources of funding (government, industry, not-for-profit, etc.) Estimate the economic impacts of this investment, using the BC Input-Output Model (BCIOM). Develop a value proposition targeting stakeholder audiences, including the relevant provincial and federal healthcare agencies. www.bccrin.ca ! w e N Invest in people through facilitating certification SoCRA Certified Clinical Research Professional program 25 Clinical Research Professionals Support 80% of certification fees (exam, membership, study guide) Study Group Recognition www.bccrin.ca Heather Harris hharris@bccrin.ca 604.675.1023 www.bccrin.ca www.bccrin.ca