Lab 3 Gases Pressure v Volume

GAS PRESSURE AND VOLUME
The purpose of this experiment is to determine the relationship between the pressure applied to a gas and the volume of the gas. To accomplish this, you will trap a constant amount of air in a sealed syringe and increase the pressure on the air by placing weight (textbooks) on the syringe plunger, recording the number of books and the corresponding air volume each time.
Procedure
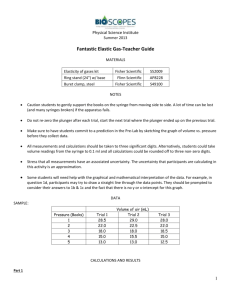
A convenient data table is shown at right.
1. Set up a sealed syringe with an air sample trapped inside and center a textbook on top of the plunger. Record the pressure (1 book) and the corresponding volume.
2. Add another book, so that 2 books rest on the plunger, and record the gas volume.
Continue in this manner until a pressure of 6 or 7 books is obtained. You may need to steady the books, but try to make all of their weight on the air sample, not leaning on the support rod.
# of books gas V (mL)
Analysis
A convenient analysis table is shown at right.
Begin your analysis with this table, and copy the information from your data table into the first two columns of the analysis table.
P (books) gas V (mL) 1/V (mL
–1
) true P (books) P true
x V (book•mL)
1. Prepare a graph of pressure exerted on the gas (# of books) vs. volume. Draw a smooth curve through the points.
2. Find and record the reciprocal volume, 1/V, for each trial. On the upper half of a new page, prepare a new graph of P vs. 1/V. Draw the best smooth curve through these points.
3. In our plot, we indicated that P = 0 when there were no books on the syringe plunger, but that is not true –– we neglected the atmospheric pressure. In fact, the point where pressure is truly zero should be shifted down to where there is neither book pressure nor atmospheric pressure on the plunger.
From your first graph, you can see that as the pressure becomes less and less, the volume increases.
As the volume become larger, the reciprocal volume (1/V) becomes smaller, until it approaches zero.
When the total pressure is truly zero, the volume is huge, so 1/V ≈ 0.
Extend your line back to 1/V = 0. The pressure at this point is truly zero. Don't worry that on your graph it appears to have a negative value; that is because we neglected to consider the atmospheric pressure in the first place. The value shown on your graph is the number of books whose weight is equivalent to the atmospheric pressure. Record this value on your graph.
4. Calculate the true total pressure for each point by adding the book equivalent of atmospheric pressure to the observed number of books. Record these values as the true pressure.
5. Find the product of true pressure x volume for each trial. Round each answer to 2 digits before recording the values in the final column of your analysis table.
Discussion
1. State what relationship you have observed between pressure and volume, in words and mathematically.
2. What variables were assumed to be constant during this experiment?
3. How do you know that the amount of air in the syringe did not change during the experiment (i.e., that the syringe did not leak)?
4. Describe at least two significant sources of error in the experiment. These are not analysis errors, but procedural problems or design flaws that interfere with obtaining accurate data. For example, making that graph too small is a mistake (because the value for the atmospheric pressure in books would be hard to determine accurately), but it is not a source of experimental error because you could draw a new graph with the same data. Supporting too much of the book weight yourself is a source of experimental error because it causes the volume readings to be inaccurate, and the only way to overcome the error is to do the experiment again.