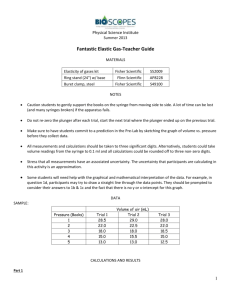
NAME: SECTION: DATE: GROUP A GROUP B 10-1 10-2 10-3 10-4 10-5 ACTIVITY 1: PUMP ME IN AND OUT INVESTIGATING BOYLE’S LAW Objectives: At the end of the lesson, you should be able to: a. Determine the relationship of Pressure and Volume b. Calculate problems relating to Boyle’s Law PLUNGER Materials: Syringes (at Least 10cc), Paper, Pen, and Observation sheet Procedure: 1. Get a 10 cc syringe and record its volume. GRADUAT BARREL Q1: what is the total volume content of the syringe? ________ TIP 2. Cover and tip of the syringe 3. while you are covering the tip of the syringe push the plunger down Q2: what happen to the volume of gas inside the barrel or cylinder?______________________________ Q3: what happen if you continue to push the plunger?_________________________________________ Q4: why does the plunger stop at a certain point?_____________________________________________ 4. while still covering the tip of the syringe release the plunger? Q5: What happen to the pressure? ________________________________________________________ Q6: Based from procedure 3 and 4. What is the relationship of PRESSURE against VOLUME and vice versa. ________________________________________________________________________________ Problem solving: (show your solutions) Q6: If we have a gas sample of 5ml at 10 atm what is the new pressure if we change the volume to 7ml?