Lewis Structure Example Problems
advertisement

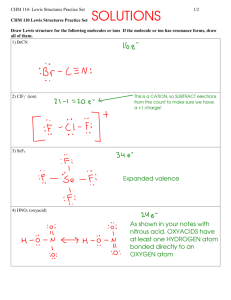
CHM 114 Topics 03/07/16 Lewis Structure Example Problems The following steps will help you to draw Lewis structures from a molecular formula. 1. Arrange the element symbols so that each atom is next to the atom(s) to which it will be bonded. This step may just be a guess. That is alright, if the subsequent steps cannot be completed then your initial placement of the atoms was wron. If this happens, then just try a different arrangement. The one pertinent hint for this first step is to place hydrogens on the outside of the molecule since they can only form one bond. 2. Count the number of valence electrons for each atom and sum these for the entire molecule. If the molecular formula represents an ion, add one more electron for each negative charge or subtract one electron for each positive charge. 3. Pair up the total number of electrons determined in step 2 and distribute them, abiding by the octet rule and maximizing the total number of bonds. Problem 1: Draw the Lewis structure of methane (CH4). Solution Problem 2: Draw the Lewis structure of acetylene (C2H2). Solution Problem 3: Draw the Lewis structure(s) for C2H4O. Solution Problem 4: Draw the Lewis structure for CH3+. Solution Problem 5: Draw the Lewis structure for CH6N+. Solution