File
advertisement

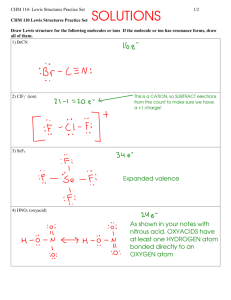
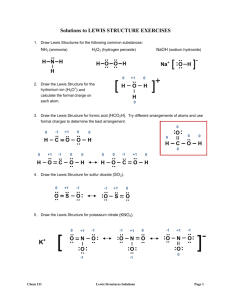
Homework 1: Advanced bonding (chapter 6) Need a separate piece of paper to complete Write the lewis dot notation for the following elements: Na Al S Xe Ba Ge P I Lewis Structures Day 1: Following the steps described in class draw a valid lewis structure for each molecule. (make sure to include all bonded and lone pairs in your drawing SiO2 PBr3 CF4 Cl2Se SiF2S N2 Lewis Structures Day 2: Following the steps described in class draw a valid lewis structure for the following. (make sure to include all bonded and lone pairs in your drawing. These molecules all have “resonance” properly show all resonant structures: NO2- SO3 N 2O Polyatomic ions are covalently bonded atoms that carry a charge, draw the correct lewis structure for each BrO- BrO4- CO32- PO33- Organic molecules CH3CH2CH3 CH2CH2 C2H2 NH4+