File
advertisement

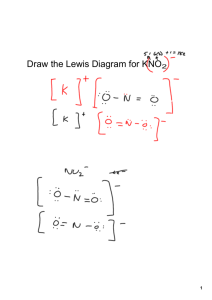
Name_______________ Ionic Compounds & Formula Writing Date___________B____ Chemists use a _________________ set of rules to write and name Ex. 1 Aluminum Nitride compounds. Every ionic compounds must be electrically ___________, meaning the total positive and negative charge is __________. It is also a Cation rule that the ___________ cation is written first and the ______________ Anion Formula = ____________ anion second (element name changed to the _______ suffix). A number in the _________ ________ of the element symbol tells us Ex. 2 Barium Chloride ______ _______ of those atoms there are in a compound. For example, _____________ means that there are ____ Na atoms and ____ O atoms. Cation Anion Formula = ____________ MONOATOMIC IONS Formula Writing Practice Problems: Anions Cations Bromide Sulfide Nitride Sodium Barium Gallium Transition have Ex. 3a Copper (II) Oxide metals Ex. 3b Chromium (III) Oxide ___________ ion charges that are shown using roman numerals. For example, ____ is called _____________. Cation Anion Formula = ____________ Cation Anion Formula = ______________ Practice Problems: Name the following using roman numerals to show charge and the “-ide” ending 1. CuCl 3. CrS 5. FeO 2. CuO 4. CoF2 6. Fe2O3 Name_______________ Ionic Compounds & Formula Writing Date___________B____ There are also many ______________________ ions (ex. Ex. 4 Sodium Phosphate ________________________________). These groups of atoms form groups with their own special names and specific charges. Polyatomic ions should be treated as a ___________ object or Cation Anion Formula = _______________ group. The subscripts of a polyatomic ion can ______ be changed. Practice Problems: Name the following using polyatomic ion names or the “-ide” ending 1. Na2CO3 3. BaSO4 5. Li3PO4 2. NH4OH 4. KNO3 Chemists use ________________ Ex. 5 6. NH4NO3 Strontium Nitrate Ex. 6 Ammonium Sulfate to show when there is more than one polyatomic ion, for example _______ means that there are Cation Anion Cation Formula = _______________ Anion Formula = ______________ ___________________. POLYATOMIC IONS Formula Writing Practice Problems: use ( parentheses ) when needed Anions Cations Potassium Ammonium Strontium Aluminum Hydroxide Nitrate Sulfate Phosphate Ionic Compounds & Formula Writing Name_______________ Date___________B____ Practice Problems - Write the formula of each compound ( Write the ions first!) 1. Cesium Fluoride Cation 5. Lithium Sulfide Anion 9. Lithium Phosphate Cation Anion Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 2. Zinc (II) Oxide 6. Copper (II) Hydroxide 10. Ammonium Chloride Cation Anion Cation Anion Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 3. Sodium Carbonate 7. Aluminum Oxide 11. Cobalt (III) Nitrite Cation Anion Cation Anion Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 4. Barium Bromide 8. Silver (I) Nitrate 12. Francium Phosphite Cation Anion Formula = _______________ Cation Anion Formula = _______________ Cation Anion Formula = _______________ MIXED REVIEW: Formula Writing with Monoatomic and Polyatomic Ions Anions Cations Lithium Magnesium Aluminum Iodide Sulfide Nitrate Carbonate Phosphate Ionic Compounds & Formula Writing Name_______________ Date___________B____ Practice Problems - Write the formula of each compound ( Write the ions first!) 1. Rubidium Iodide Cation 4. Ammonium Hydroxide Anion Cation Anion 7. Magnesium Nitrate Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 2. Gold (I) Chloride 5. Lead (II) Sulfide 8. Ammonium Carbonate Cation Anion Cation Anion Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 3. Barium Nitrate 6. Aluminum Sulfate 9. Silver (I) Cyanide Cation Anion Cation Formula = _______________ Anion Formula = _______________ Cation Anion Formula = _______________ Name each of the following compounds: ** These examples have transition metals and need roman numerals in their names to show charge 1. CsF 2. MnCl2 6. Cu2SO4 ** 3. Al2S3 4. Ni(OH)2 5. (NH4)3P ** ** 11. Al(NO3)3 7. NH4Cl 12. FePO4 ** 8. K3PO4 13. Fe3(PO4)2 ** 9. CsNO3 14. NaOH 10. CuCO3 ** 15. (NH4)3PO4 Ionic Compounds & Formula Writing Name_______________ Date___________B____ MORE MIXED REVIEW: Formula Writing with Monoatomic and Polyatomic Ions Anions Fluoride Oxide Hydroxide Sulfate Phosphite (not Phosphate!) Cations Cesium Ammonium Strontium Indium MORE MIXED REVIEW: Formula Writing with Monoatomic and Polyatomic Ions Anions Cations Lithium Indium Magnesium Ammonium Phosphide Sulfite (-ide, not -ate or -ite) (-ite, not -ate or -ide) Permanganate Chlorate Ionic Compounds & Formula Writing Name_______________ Date___________B____ MORE MIXED REVIEW: Formula Writing with Monoatomic and Polyatomic Ions Anions Cations Fluoride Phosphite Oxide Acetate Chromate Aluminum Cesium Ammonium Calcium Practice Problems - Write the formula of each compound ( Write the ions first!) 1. Gallium Sulfide Cation 4. Ammonium Sulfate Anion Cation Anion 7. Barium Hydroxide Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 2. Gold (I) Cyanide 5. Lead (II) Nitrate 8. Ammonium Phosphate Cation Anion Cation Anion Cation Anion Formula = _______________ Formula = _______________ Formula = _______________ 3. Calcium Sulfate 6. Nickel (II) Hydroxide 9. Silver (I) Carbonate Cation Anion Formula = _______________ Cation Anion Formula = _______________ Cation Anion Formula = _______________