Chapter 12 Review
advertisement

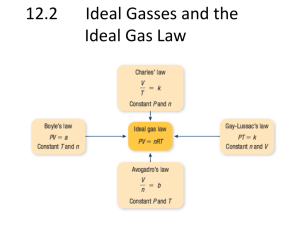
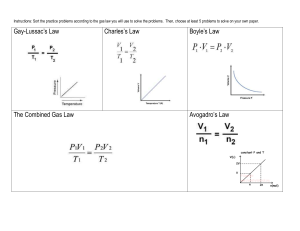
Gas Laws Review 1. What are the 4 variables for a gas along with their units? 2. What are the equations for Charles’, Boyle’s, Gay-Lussac’s ? 3. A metal cylinder contains 1 mol of gas at STP. What will happen to the pressure if another mole of gas is added to the cylinder at STP, but the temperature and volume remain constant. 4. A gas with a volume of 4 L is allowed to expand to a volume of 12 L. What happens to the pressure in the container if the temperature remains constant? 5. Describe an ideal gas. 6. A gas with a volume of 4.0 L at a pressure of .90 atm is allowed to expand until the pressure drops to .20 atm. What is the new volume? 7. A gas is compressed at a constant temperature from 27 L to 3.0 L. If the initial pressure is .50 atm, what is the final pressure? 8. A ball has a volume of 4.00 L and a pressure of 100 kPa. What is the volume of the ball if the pressure is increased to 135 kPa? 9. Use Gay-Lussac’s Law to explain the danger of throwing an aerosol can into the fire? 10. A nitrogen molecule travels at 500 m/s at room temperature. What is the velocity of a helium molecule at the same temperature? 11. An unknown gas effuses through an opening at a rate of 1.59 times slower than nitrogen gas. What is the molecular mass of the unknown gas? 12. The barometer at an indoor pool read 105.00 kPa. If the temperature of the room is 30.0 0C and the pressure of the water is 4.25 kPa, what is the partial pressure of the dry gas? 13. What is the definition of pressure? 14. What relationship does Boyle’s Law express? 15. What relationship does Charles’ Law express? 16. What relationship does Gay-Lussac’s Law express? 17. What is effusion and diffusion? 18. How does wind illustrate that gasses are fluid? 19. What are the properties of gasses? 20. What are the properties of an ideal gas? 21. 1 mole of any gas will occupy what volume? 22. Ammonia, NH3 and alcohol C2H5OH are released back into the room. Which will you smell first? (lighter gasses move faster) 23. A gas sample has a volume of 125 mL at 91.0 kPa. What is the new volume at 101 kPa? 24. The pressure of a 240.0 mL sample of helium is .428 atm. What is the new volume if the temp. drops to 295 K? 25. A balloon is filled with 2.3 L of helium at a temperature of 311 K. What is the new volume if the temp. drops to 295 K? 26. A sample of hydrogen exerts a pressure of .329 atm at 47 degrees Celsius. What will the pressure be at 77 degrees Celsius? 27. How many moles of air are in 1.00 L at -23 degrees Celsius and 101 kPa? 28. A plastic weather balloon contains 65 L of helium at 20 Celsius and 94 kPa. What is the mass of helium in the balloon? 29. What will the new volume of the balloon from 28 be at -61 degrees Celsius and 1.1 kPa? 30. How many liters of hydrogen gas can be produced at 300 K and 104 kPa if 20 g of sodium is placed in water? Na + H2O → NaOH + H2 31. What mass of magnesium will react with .500 L of oxygen at 150 degrees Celsius and 70 kPa? Mg + O2 → MgO