Gas Law Quiz
advertisement

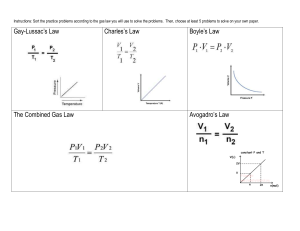
Gas Law Quiz Pd 8 Question 1 • A high altitude balloon contains 2.50 L of helium gas at 105 kPa. What is the volume when the balloon rises to an altitude where the pressure is only 40.5 kPa? Assume temperature remains constant. Question 2 • A balloon inflated in a room at 325 ºC has a volume of 6.80 L. The balloon is then cooled to a temperature of 25.0 ºC. What is the new volume if the pressure remains constant? Question 3 • The gas left in a used aerosol can is at a pressure of 103 kPa at 25 ºC. If this can is thrown onto a fire, what is the pressure of the gas when its temperature reaches 928 ºC? Assume the volume of the can remains constant. Question 4 • The volume of a gas-filled balloon is 30.0L at 40.0 ºC and 153 kPa. What is the new volume if the balloon reaches STP? Question 5 • Go back to the first four questions and give the name of the specific gas law. Answers 1. 2. 3. 4. 6.48 L 3.39 L 415 kPa 39.5 L Boyles Charles Gay-Lussac Combined