Boyle's Law Sample Laboratory Data
advertisement

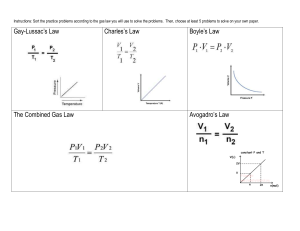
Boyle’s Law Sample Laboratory Data You probably started experimenting with Boyle's Law when you were a small child. When you squeeze a balloon, you might notice that the harder you push, the harder it seems to push back. When you lie back on an inflatable mattress, or pool float, it compresses up to a point and then seems to stop. This is because as you decrease the volume of a confined gas, the pressure that it exerts increases. This relationship, called Boyle's Law, is summarized by the statement: The volume of a sample of gas is inversely proportional to its pressure, if temperature remains constant. Use the sample data in the table below to plot and graph and answer the questions associated with Boyle’s Law. Pressure is the y variable and volume is the x variable. Trial Pressure (kPa) Volume (mL) 1 100 kPa 40 mL 2 50 kPa 80 mL 3 200 kPa 20 mL 4 400 kPa 10 mL 5 25 kPa 160 mL Questions: 1. What does the data show happens to the pressure when the volume is double? 2. What does the data show happens to the pressure when the volume is halved? 3. What does the data show happens to the pressure when the volume is tripled? 4. Does the data and the shape of your plot support that pressure and volume are inversely related? How? 5. Based upon your graph, what would you expect the pressure to be if the volume was increased to 600 mL? 6. Based upon your graph, what would you expect the volume to be if the pressure is changed to 60 kPa? Boyle’s Law Calculations: 1. A sample of gas has a volume of 33.2 mL and a pressure of 105 kPa. What will the new pressure be if the volume expands to 75 mL? 2. A balloon contains 1.23 L of air at 101.3 kPa (standard pressure). What will the volume of air in the balloon be if the pressure decreases to 64.5 kPa? 3. What is the new volume of a gas whose original volume is 555 mL? The original pressure is 120 kPa and it is increased to 145 kPa. 4. What is the new pressure of a gas whose volume is changed to 200 mL? The starting conditions were a pressure of 175 kPa and a volume of 110 mL. 5. A balloon is filled with 30 L of helium at 101.3 kPa. What is the volume when the balloon rises to an altitude where the pressure is only 25 kPa? Boyle’s Law Concept Review: 1. What two variables are involved in Boyle’s Law? 2. What conditions must remain constant in order for Boyle’s Law to be valid? 3. What is the relationship between the variables in Boyle’s Law – direct or inverse? What does this mean? 4. What happens to the volume if the pressure on a gas is doubled? Answer the following questions based upon the graph below: 5. What is the pressure is associated with a volume of 200 mL? 6. What is the volume associated with a pressure of 100 kPa? 7. What would be the volume of gas if the pressure is increased to 1000 kPa? 8. What would be the pressure of the gas if the volume is decreased to 0.2 mL? Charles’ Law Sample Laboratory Data An inflatable pool float may seem quite firm as it sits on a deck in the hot sun. However, minutes after you toss to float into the cold pool, the same float may seem under-inflated. You may suspect that the float has developed a slow leak, but that may not be the most likely explanation for the apparent loss of air pressure. It may be that Charles's law is responsible. Use the sample data in the table below to plot and graph and answer the questions associated with Charles’ Law. Graph volume (L) as the y variable and temperature (K) as the x variable. Remember…temperatures must be converted to Kelvin values! (K=C+273) Trial Volume (L) Temperature (C) Temperature (K) 1 1.0 L 27 C 2 1.33 L 127 C 3 2.0 L 327 C 4 2.33 L 427 C 5 3.0 L 627 C Questions: 1. What does the data show happens to the volume as the temperature is doubled? 2. What does the data show happens to the volume as the temperature is halved? 3. What does the data show happens to the volume as the temperature is tripled? 4. According to the data and the plot, what is the relationship (direct or inverse) between volume and temperature as described by Charles’ Law? What evidence supports this? 5. Based upon your graph, what would you expect the volume to be if the temperature is raised to 1000 K? 6. Based upon your graph, what would you expect the volume to be if the temperature is decreased to 100 K? Charles’ Law Calculations: 1. If 5 mL of carbon dioxide gas at -140 C expands to a volume of 15 mL, what is the new temperature of the gas? 2. A balloon inflated in an air conditioned room at 27 C has a volume of 4.0 L. The balloon is taken outside where the temperature raises up to 57 C. What is the new volume of the balloon? 3. If a sample of gas occupies 6.8 L at 75 C, wha twill be its volume at 35 C? 4. What is the new temperature of a gas whose volume is 20 L? The original temperature was 7 C and the original volume was 12 L. 5. Find the new volume of a gas when 90 mL of it is heated from 27 C up to 42 C. Gay-Lussac’s Law Calculations: 1. A gas has a pressure of 50 kPa at 540 K. What will be the new pressure at 200 K if the volume remains constant? 2. The pressure of a sample of gas in a gas cylinder is 300 kPa at standard temperature (0 C). What pressure would the gas exert at a temperature of 100 C? 3. An ideal gas exerts a pressure of 105 kPa at 273 K. At what temperature would this gas exert a pressure of only 30 kPa? 4. A tire is inflated to 110 kPa at 50 C. What will be the new pressure if the temperature increases to 85 C after driving? 5. A gas exerts a pressure of 75 kPa when at 33 C. At what temperature will the pressure equal 140 kPa? Combined Gas Law Calculations: 1. A container with an initial volume of 1.0 L is occupied by a gas at a pressure of 150 kPa and a temperature of 25 C. What is the new volume required to hold the gas if the pressure changes to 225 kPa and the temperature changes to 100 C? 2. A volume of 500 mL of gas is at 120 kPa and 27 C. Find the new pressure if the volume is decreased to 350 mL and the temperature decreases to -3 C. 3. A container holds 800 mL of a gas at a pressure of 200 kPa and a temperature of 30 C. Determine the new volume when the conditions of pressure and temperature are brought to standard values (101.3 kPa, 0 C). 4. If 100 mL of gas are at 65 kPa and 17 C, find the new volume at 95 kPa and 145 C. 5. A 60 L sample of gas is held at standard values of temperature and pressure. What will be the new temperature if the gas is compressed to 32.5 L under a pressure of 320 kPa? The Gas Laws – Review Questions and Calculations: 1. There are four variables that impact the behavior of gases. What are they? 2. Which of the gas laws discussed reflect a direct relationship among the variables? 3. Which of the gas laws discussed reflect an inverse relationship among the variables? 4. Which variables must remain constant in order for Charles’ Law to be accurate? 5. Which variables must remain constant in order for Gay-Lussac’s Law to be accurate? 6. Which variable must remain constant in order for the Combined Gas Law to be accurate? 7. How would you expect pressure to change if the volume of the gas is doubled? 8. How would you expect the volume of a gas to change if the temperature is cut in half? 9. How would you predict the temperature to change if the pressure of the gas is tripled? 10. What would be the new pressure of a gas at 100 kPa if the volume is changed from 25 mL to 65 mL? 11. A gas at 45 C is heated up to 100 C. What will be the new pressure if the original pressure was 95 kPa? 12. A 3 L volume of gas is cooled from 125 C down to 110 C. What is the new volume? 13. At a pressure of 110 kPa, 200 mL of gas is held at 30 C. What will be the new pressure of the gas if the volume is decreased to 150 mL and the temperature increases to 75 C? Moles of Carbon Dioxide from Antacid Tablets Purpose: To measure the amount of carbon dioxide gas given off when antacid tablets dissolve in water. Materials: 6 effervescent antacid tablets 3 spherical rubber balloons Plastic medicine dropper Water Metric tape measure Graph paper Funnel Procedure: 1. Break one antacid tablet into small pieces. Using the funnel, put the broken pieces into a balloon. 2. Add 5 mL of water to the balloon and immediately tie off the balloon. 3. Shake the balloon to mix the contents. 4. Measure and record the circumference of balloon several times during the next 20 minutes. Determine the maximum circumference achieved. 5. While waiting, break up two more antacid tablets into small pieces and dump them into another balloon. 6. Add 5 mL of water to this balloon and immediately tie it off. 7. Shake the balloon to mix the contents. 8. Measure and record the circumference of balloon several times during the next 20 minutes. Determine the maximum circumference achieved. 9. While waiting, break up three more antacid tablets into small pieces and dump them into a third balloon. 10. Add 5 mL of water to this balloon and tie off immediately. 11. Shake the balloon to mix the contents. 12. Measure and record the circumference of balloon several times during the next 20 minutes. Determine the maximum circumference achieved. 13. Use the maximum circumference recorded for each balloon to calculate the volume achieved by each balloon. (Formulas: Volume = 4πr3/3 and r = circumference/2π) Conclusion: 1. Plot a graph of volume versus number of tablets. Use your graph to describe the relationship between the number of tablets used and the volume of the balloon. 2. Assume that the balloon is filled with carbon dioxide gas at 20°C and standard pressure. Calculate the mass and the number of moles of CO2 in each balloon at maximum inflation. (Hint: Use the Ideal Gas Law to solve) How many moles of CO2 did each antacid tablet produce? 3. If a typical antacid tablet contains 2.0 grams of sodium hydrogen carbonate, how many moles of CO2 should one tablet yield? Compare this theoretical yield with your actual results. (Use a 1:1 mole ratio.) Review of Mole-Mass and Mass-Mole Conversions: 1. How many moles of neon are equal to 52.5 grams of the gas? 2. How many grams of iron are in 2.5 moles of the metal? 3. What is the molar mass of MgSO4? 4. How many moles of H2O are produced if the mass produced is measure to be 28.5 grams? 5. How many grams of NaCl should be produced from a reaction aimed at producing 3 moles of the salt? Ideal Gas Law Calculations: 1. Calculate the pressure of 1.65 g of helium gas at 16°C. The gas occupies a volume of 3.25 L. 2. What will be the volume of 0.70 g of N2 gas at 0°C and 150 kPa of pressure? 3. How many moles of Ar are in a 27.3 L sample of the gas at a pressure of 85.5 kPa and a temperature of 50°C? 4. What volume will 50 grams of Cl2 occupy at 0°C and 101.3 kPa? 5. How many grams of gas are found in a 5 L container if the conditions inside are 25°C and 200 kPa? Solubility Varies with Temperature: 1. What is the solubility of KNO3 at 10°C? 2. How many grams of KBr can be dissolved (in 100 grams of H2O) at 80°C? 3. Which one of the salts on the graph is the most soluble at 20°C? 4. Which one of the salts on the graph is the least soluble at 10°C? 5. How many grams of NaCl can be dissolved (in 100 grams of H2O) at 70°C? 6. If 40 grams of NaNO3 is added to 100 grams of H2O at 20°C, will the solution be saturated, unsaturated, or supersaturated? 7. If 80 grams of NH4Cl is added to 100 grams of H2O at 60°C, will the solution be saturated, unsaturated, or supersaturated? 8. If 78 grams of KBr is added to 100 grams of H2O at 40°C, will the solution be saturated, unsaturated, or supersaturated? 9. How many grams of KNO3 will precipitate out of solution if the temperature is decreased from 70°C down to 40°C? 10. How much additional NaNO3 must be added to solution to maintain saturation as the temperature is increased from 30°C up to 80°C? REVIEW: 1. What type of bond would form between an atom of sodium and an atom of chlorine? Why? 2. What type of bond would form between atoms of oxygen and fluorine? Why? 3. Draw the diagram for a molecule of COCl2. Use Lewis dot diagrams to determine bonding. 4. What is the name of the molecular shape formed in question #3? 5. Is the molecule in question #3 polar or nonpolar? 6. Draw the diagram for a molecule of CO2. Use Lewis dot diagrams to determine bonding. 7. What is the shape of the molecule in question #6? 8. Is the molecule in question #6 polar or nonpolar? 9. Draw diagrams for the two possible isomers of C4H10. 10. Determine the name of each of the following organic molecules: 11. Draw diagrams for each of the following organic molecules: a. 3 ethyl 2 methyl pentane b. 2 methyl 4 propyl 3 heptene 12. Which of the gas laws describe an inverse relationship between the variables? What does this mean? 13. Which of the gas laws describe a direct relationship between the variables? What does this mean? 14. A 2 L sample of gas is held at 25° and 101 kPa of pressure. What will be the new volume if the temperature increases to 50°C and the pressure increases to 120 kPa? 15. How many grams of CO2 are in a 2.5 L container held at 40°C and 120 kPa of pressure? Unit 10: The Gas Laws I. A Comparison of Solids, Liquids, and Gases 1. Diagram: 2. Phase Changes: a. melting – b. freezing – c. evaporation – d. condensation – e. sublimation – f. deposition – 3. Compare and Contrast: II. The Kinetic Molecular Theory (5 statements): 1. 2. 3. 4. 5. III. Pressure and Temperature Conversions: 1. Definition of Pressure: 2. Values of Standard Pressure: (convert using conversion factors) 3. Values of Standard Temperature: (convert using the formula) IV. Boyle’s Law: 1. Definition 2. Formulas: Sample Calculation: A 380.3 mL volume of gas is at a pressure of 750 torr. What would be the new volume if the pressure is changed to 790.8 torr? V. Charles’ Law: 1. Definition 2. Formulas: Sample Calculation: What would be the new volume of gas if a 500 mL quantity at 173 K is cooled to 0 C? VI. Gay Lussac’s Law: 1. Definition 2. Formulas: Sample Calculation: The gas in an aerosol can is at a pressure of 1 atm and a temperature of 27 C. What would the new pressure be if the temperature is raised to 100 C? VII. The Combined Gas Law: 1. Definition 2. Formulas: Sample Calculation: A 30 L cylinder of oxygen is at a pressure of 100 atm and a temperature of 27 C. What would be the new temperature if the volume is decreased to 20 L and the pressure to 5 atm? Homework Handout: Pressure and Temperature Conversions Part I: Pressure Conversions – Convert the following pressures using the conversion factor method. 1. 18.4 atm = ____________mm Hg 2. 125.2 kPa = ____________torr 3. 11.1 psi = ____________atm 4. 731 torr = ____________psi 5. 797 mm Hg = ____________kPa 6. 98.2 kPa = ____________torr Part II: Temperature Conversions – Convert the following Celsius temperatures to Kelvin. 1. 50 C = ____________K 2. 125 C = ____________K 3. 72 C = ____________K 4. 25 C = ____________K 5. -15 C = ____________K 6. -35 C = ____________K Boyle’s and Charles’ Law Review: 1. A sample of gas has a volume of 45.9 mL and a pressure of 770 mm Hg. What will the new pressure be if the volume expands to 75 mL? 2. If 15.2 mL of carbon dioxide gas at 10.5 C expands to a volume of 35.5 mL, what is the new temperature of the gas? 3. A balloon contains 2.40 L of air at 101.3 kPa (standard pressure). What will the volume of air in the balloon be if the pressure decreases to 12.5 psi? 4. A balloon inflated in an air conditioned room at 32 C has a volume of 5.25 L. The balloon is taken outside where the temperature raises up to 65 C. What is the new volume of the balloon? 5. What is the new volume of a gas whose original volume is 350 mL? The original pressure is 745 torr and it is increased to 145 kPa. 6. If a sample of gas occupies 125 mL at 75 C, what will be its volume at 35 C? 7. What is the new pressure of a gas whose volume is changed to 135 mL? The starting conditions were a pressure of 1.20 atm and a volume of 110 mL. 8. What is the new temperature of a gas whose volume is 367 mL? The original temperature was 25.5 C and the original volume was 525 mL. 9. A balloon is filled with 12.5 L of helium at 760 torr. What is the volume when the balloon rises to an altitude where the pressure is only 75.5 kPa? 10. Find the new volume of a gas when 145 mL of it is heated from 75.0 C up to 125 C.