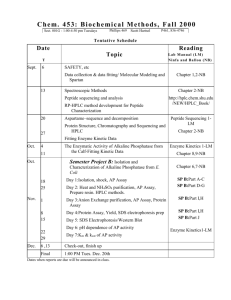
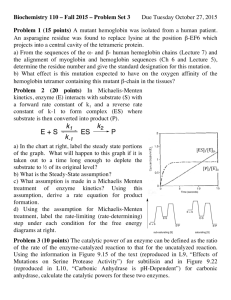
C582/B504 Review Problem Set 1

Review Problem Set 3
Skills questions:
1) Draw a notional active site for alkaline phosphatase. Define roles for each of the residues. Draw a mechanism for the enzyme-catalyzed reaction. Replacement of the active site serine with alanine gives an enzyme with altered catalytic properties. Predict the effects on Km and kcat. How would you explain low levels of residual enzyme activity?
2) Delineate the major groups of phosphatases. List whether they use an active site nucleophile and/or metal ions.
3) To trap a phosphoenzyme intermediate, what must be true about the relative rates of the microscopic (step by step) rate constants. Draw a kinetic scheme and free energy profiles to illustrate this.
4) Inorganic phosphate is a potent inhibitor of alkaline phosphatase. Under some conditions, it binds so tightly that dissociation of Pi is the rate limiting step. Draw a free energy profile for this process and comment about this phenomenon in terms of differential binding and optimization of the catalytic process. Why might this adaptation be useful? How might you speed up this enzyme (in general terms)?
5) Outline how a protein may be synthesized by native chemical ligation strategies.
What are the major limitations on this methodology? What major advantage does this process have vs. site-directed mutagenesis for understanding enzyme function?
6) If you haven’t done so yet, please read the 1993 Kent paper in PNAS. Diagram HIV protease and show what Kent was probing? What do they find and how do they explain this? Explain their inhibitor experiments. Do you find them convincing?
7) Outline the major structural motifs available for the design of transition state analogs that will inhibit aspartyl proteases. (You may find a summary in the Wong review.)
Explain why these structures would be expected to be tight binding inhibitors. Which of these is being used in the inhibitor structures shown in figure 11?
8) What is a Bronsted plot? Explain what such a plot is designed to show. Graph out a notional reaction of a series of aromatic phosphate esters with a beta value of -1. What is the interpretation of this? Alkaline phosphatase shows a beta value for kcat of close to zero. What possible interpretations are there of this result? Which do you think is correct?