Collision Theory Worksheet: Chemistry Kinetics
advertisement

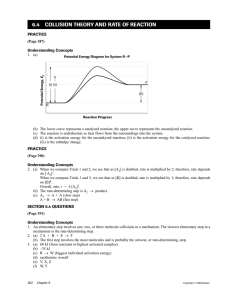
Collision Theory Questions CHE 40S Part I. Collision Theory 1. What 2 criteria must be met in order for a reaction to proceed towards the formation of products? Particles must collide (collision frequency) with the appropriate geometry and sufficient energy to produce an activated complex (fraction of effective collisions). 2. Explain using collision theory how each of the following observations affect the rate of a chemical reaction. Use diagrams where necessary to support your reasoning. a) The concentration of a reactant molecule is increased. More molecules mean that more collisions will occur in the same given space, increasing the number of successful collisions. There will be a greater number of molecules with sufficient activation energy to form an activated complex. Reaction rate will increase. A A A A B B 1 possible collision 5 possible collisions b) A catalyst is added. Reaction rate will increase because a catalyst provides an alternate, lowerenergy pathway for the formation of a different activated complex. Potential Energy Uncatalyzed reaction Catalyzed reaction Reaction Pathway c) The surface area of a solid reactant is decreased. A lower surface area means that less reactant molecules are available to react, therefore, decreasing the frequency of collisions and lowering the reaction rate. 3. a) Why must a match be struck in order for it to burn, and A match will not burn unless the activation energy barrier is overcome as occurs when a match is struck. b) why does it burn all by itself after being struck? A match burns to completion after being struck because the molecules of wood have sufficient stored or potential energy to keep the combustion reaction going. Part II. Kinetics Terms 4. Define or explain the following terms. a) activated complex An activated complex is a high-energy, short-lived, intermediate molecule formed at the peak of the activation energy barrier. b) change in enthalpy Is the difference in energy between the reactant particles and the product particles in a chemical reaction. The change in enthalpy can be negative or exothermic, indicating heat is released during the reaction, or the change can be positive, indicating an endothermic reaction requiring a net input of energy.