SCH4U Exam Review: Organic, Structure and Electro Chemistry
advertisement

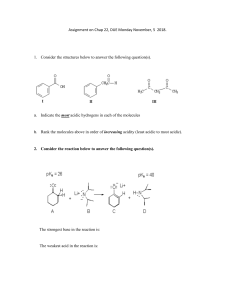
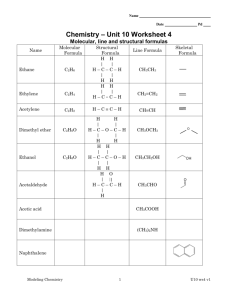
SCH4U Exam Review: Organic, Structure and Electro Chemistry Organic Chem: Know all functional groups & naming/drawing rules Know important reactions: Halogenation (with Br2, Cl2) Hydrogenation (with H2) Hydrohalonation (with HBr, HCl) Hydration (with H2O) Preparation: Hydrolysis: If an ester is reacted with an acid or base the ester will split into it’s alcohol and carboxylic acid Markovnikov’s Rule: When a hydrogen halide or water is added to an alkene or alkyne the hydrogen atom will bond to the carbon atom within the multiple bond that already has more hydrogen atoms. Structure Chemistry: The Aufbau principle tells us that when building energy diagrams we always start at the lowest energy level and then build up No two electrons can be put into the same orbital of equal energy until one electron has been put into each of the equal energy orbital – this is Hund’s Rule Electrons are spinning, they always go in opposite directions – this is known as the Pauli Exclusion Principle. Energy Level diagrams Draw the energy level diagram and electron configuration for: copper nitrogen ion VSEPR Theory: o Know how to use the table! What are the shapes of the following molecules: BF3 CCl4 NH3 SF6 PF3 CO2 Electro Chemistry: LEO the lion says GER NO2¯(aq) + Al(s) NH3(g) + AlO2¯(aq) (acidic solution, oxidation # method) Cr2O72-(aq) + Cl-(aq) method) Cr3+(aq) + Cl2 (g) CN-(aq) + MnO4-(aq) CNO-(aq) + MnO2 (s) (acidic solution, half-reaction (basic solution)