V. Summary and tables of chemical shifts
advertisement

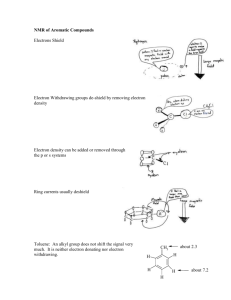
03/06/16 SCHM 530, Chapter 3 Chapter 3 Chemical Shift I. Factors that influence proton shifts A. Perturbations of electron density at H1 1. Electrons also precess in Bo a) Lenz's law - magnetic field that arises from electron precession opposes the direction of Bo b) "The direction of an induced current is such as to oppose the cause producing it" c) Cause of shielding & chemical shift d) Diamagnetic - any material such as electron cloud that gives rise to a magnetic field that opposes Bo e) diamagnetism - the property that certain substances have of being repelled by both poles of a magnet & hence taking a position at right angles to the magnet's line of influence (from the dictionary) f) Therefore "diamagnetic shielding" sigmad spherically symmetric circulation of the electrons because of the structure of the molecules (1) Proportional to electron density around the nucleus g) Maximal effect - precession unhindered as when electron cloud spherical (1) Free atoms (2) Some molecules with tetrahedral symmetry (a) Methane, ammonium ion 1 03/06/16 SCHM 530, Chapter 3 2. Proton dominated primarily by diamagnetic shielding 3. Therefore magnetic field increased or frequency decreased to compensate for shielding 4. Diamagnetic shielding depend on electron density a) Electronegative element decreases sigmad b) Therefore shift to lower field (higher frequency) (1) Methane 0.2 (2) Methyl chloride 2.7 (3) Methylene chloride 5.3 (4) Chloroform 7.3 c) Nitro, CN, C=O, F, RO, OH, NH2 d) Electropositive R-Si, R-Mg 5. Electron density also dependent on resonance a) CH2=CH-OCH3 <-> -CH2-CH=OCH3+ (1) Therefore higher field (4.1) than CH2=CH2 (5.28) b) Phenyl-OCH3 - resonance with + on O and - on para (1) Therefore higher field for para H than in benzene c) Also ethyl trans-crotonate, fig 3.2, p38 6. Hybridization SP3 25%, SP2 33%, SP 50% B. Magnetic Properties of substituents 1. Spherical or isotopic substituent - no perturbing effect a) Isotopic substituent - 2 orientations cancel each other out (1) Molecule tumbling freely 2 03/06/16 SCHM 530, Chapter 3 3 2. Ellipsoidal or cylindrical substituent - major effect a) Diamagnetic anisotropy (1) Effect not averaged to 0 by tumbling b) Ellipsoidal shape electron clouds (1) Oblate (dish-like) (a) Benzene ring (b) Oblate ellipsoid - 2 extreme arrangements (i) Flat portion of ring perpendicular to Bo (a) H1 lower field because in reinforced region field (b) H1 over ring at higher field because it is in the opposed region of the field (ii) If plane of ring rotated so it is parallel to field (a) Little or no induced circulation of current (b) For benzene ring, induced field particularly large (i) Aromatic electrons circulate easily (c) Examples (i) methano [10]annulene sigma -0.5, figure 3-3 (ii) [18]annulene 3-4 c) prolate (rod-like) (1) Single or triple bonds (a) Prolate ellipsoid (b) If diamagnetic susceptibility greatest when long axis parallel (i) H1 at end shielded 03/06/16 SCHM 530, Chapter 3 4 (ii) H1 at side deshielded (c) If reverse, reverse is true (i) good model for chemical bond (d) Acetylene - pi electrons cylindrically arranged (i) Acetylene between ethane & ethylene (ii) Also true for C#N (iii) At 55o 44' effect is null (iv) Similar null point for benzene ring (e) Circulation of charge within single bond less effective than C#C (f) Cyclohexane as example (i) H1 along side more shielded than along end (ii) Axial - shielding region of 2,3 & 5,6 bonds (iii) Equatorial - deshielding region (a) Therefore Hax at 0.5 ppm higher than Heq (g) CH3X Vs CH3CH2X Vs (CH3)2CHX (i) Due to anisotropy of additional C-C bonds (h) Cyclopropane special because CH2 directly opposite C-C (i) Therefore shielded relative to regular CH2 (2) Anisotropy of C=C more difficult to assess because 3 nonequivalent axes (a) H1 over C=C shielded (i) Norbornene with H above C=C shielded (b) The aldehyde proton is severely deshielded – downfield 03/06/16 SCHM 530, Chapter 3 (c) -OH in hydrogen bonding – downfield (i) EtOH free OH at 0.7, neat at 5.3 delta (ii) H bonding lone pair - prolate ellipsoid (iii) H at end of ellipsoid & deshielded 3. Summary: most H1 chemical shifts explained by a) Electron withdrawal or donation by induction, resonance, hybridization b) Diamagnetic anisotropy - H1 in vicinity of nonspherical substituents (1) Anisotropy - exhibiting properties with different values when measured along axes in different directions (dictionary) 5 03/06/16 SCHM 530, Chapter 3 II. .Chemical Shift & Structure for protons A. Saturated alkanes 1. Cyclopropane - anisotropy of sigma bond, delta of 0.22 2. Progressive addition - downfield shift for cyclic & acyclic 3. Spin-spin splitting - broad overlapping resonances B. Functionalized Saturated Alkanes 1. Electronegativity & diamagnetic anisotropy a) O > N > S b) F > Cl > Br > I c) Multiple substituents d) -CN, -NO2, -Si C. Unsaturated Aliphatics 1. Alkynes a) Anisotropy - relatively high-field position 2. Alkenes a) Electronegativity of sp2 C b) Angle strain on C=C - move to lower fields (1) Example - norbornene CH=CH at 5.94 (2) 1,3-cyclohexadiene at 5.78 due to conjugation (3) 1,3-cyclopentadiene at 6.42 due to conjugation & strain (4) Styrene CH at 6.66, CH2 at 5.15-5.63 (a) Benzene ring is electron withdrawing 6 03/06/16 SCHM 530, Chapter 3 c) Carbonyl electron withdrawing by induction & resonance (1) Cyclohexene at 5.59 (2) Substitute O at allylic position (a) CH closest at 6.37, other CH at 4.65 (b) Resonance with + on O & - on C (3) 2-cyclohexenone (a) CH next to C=O at 5.93 (b) CH further away at 6.88 (c) Resonance with - on O & + on C 7 03/06/16 SCHM 530, Chapter 3 3. Aldehydes R-CHO at 9.80 a) In deshielding zone of anisotropic C=O D. Aromatic Molecules 1. Diamagnetic anisotropy of benzene 7.27 2. Nitrobenzene a) Ortho CH 8.22 b) Meta CH 7.48 c) Para CH 7.61 d) Resonance with - on O, + on ortho or para 3. Anisole (methoxybenzene) a) Ortho at 6.79 b) Meta at 7.24 c) Para at 6.73 d) Resonance with + on O, - on ortho or para 4. Pyridine a) Ortho 8.50 b) Meta 6.99 5. Pyrole a) Ortho 6.68 b) Para 6.22 c) Resonance with + on N, - on ortho E. proton on oxygen and nitrogen 1. -OH, -NH - acidity & H-bonding effect 8 03/06/16 SCHM 530, Chapter 3 9 a) Variable b) D2O - aqueous layer separate out c) R-CO2H - H-bonding dimers or oligomers 10-13 delta III. Factors that influence carbon shifts A. Diamagnetic shielding ( d) maximal when electron cloud is spherically symmetrical 1. Electron precession in magnetic field impeded when cloud is nonspherical 2. H1 only s electrons, spherical symmetry a) Therefore diamagnetic term dominant B. Paramagnetic term ( p) 1. Hindrance to free electronic rotation a) 2p electrons have angular momentum that hinders free rotation b) Molecules with unsymmetrical electron distributions – atoms other than H 2. Opposite direction to d 3. Paramagnetic because opposite to diamagnetic a) Don’t confuse with usage to describe molecules with unpaired electrons 4. Paramagnetic term can be quite large a) d parts/million b) p 100s/million or 1000s/million 5. Therefore range of chemical shifts extremely large 03/06/16 SCHM 530, Chapter 3 6. Sigmap p proportional to 1/E * <r-3+> * Qij a) E - average excitation energy (1) Accessibility of excited states b) <r-3> - radial term (1) Distance of electron cloud c) Qij molecular orbital description of bonding (1) Multiple bonding 10 03/06/16 SCHM 530, Chapter 3 11 C. E difference between ground & excited states 1. Average over all available excited states 2. The lower the energy of the excited state, the better able to provide shielding 3. p proportional to 1/E 4. Saturated molecules - no low-lying excited states a) Therefore at very high field (small p) b) C-alkanes c) N-amines d) O-ethers 5. Remember that paramagnetic shielding causes downfield shifts 6. C=O - opposite extreme a) n -> * b) C13 at 200 ppm downfield of TMS c) Even larger for carbocation – 335 d) N15 in nitroso N=O 900 ppm downfield of NH3 7. -> * too localized a) No significant paramagnetic current b) Therefore C=C no significant effect on E 8. Complete understanding require appreciation of molecular orbital structure D. <r-3> - average of inverse cube of distance r between nucleus & bonding electrons (especially 2p electrons) 1. Electronic location cannot be specified 03/06/16 SCHM 530, Chapter 3 a) therefore average 2. sigmap proportional to 1/r3 a) Therefore the further the electrons, the less important the paramagnetic shielding 3. Electrons closer to nucleus going left to right in periodic table (row) a) O closer 2p electrons than C (1) Therefore 17O shift = 3 times 13C shift (2) Therefore 17O more sensitive to different factors than 13C 4. Radial effect similar to electronegativity in d a) When electrons donated to C, electrons around C must move further away to reduce electrostatic repulsion, thereby increasing r & decreasing p (1) Upfield shift b) Electron withdrawing - reverse effect (1) Carbon tetrachloride 97 PPM (2) Chloroform 78 PPM (3) Methylene chloride 54 PPM (4) Methyl chloride 25 PPM 12 03/06/16 SCHM 530, Chapter 3 13 c) Numbers larger than H1 because p > d 5. Therefore substituent effect follow electronegativity E. Qij related to charge density & bond orders 1. a measure of multiple bonding a) Greater multiple bonding -> larger p -> lower field 2. Examples a) Ethane 6 b) Ethylene 123 c) Allene 214 d) Benzene 129 e) Acetylene at 70 & other alkynes more complicated (1) linear structure has zero angular momentum around the C#C axis 03/06/16 SCHM 530, Chapter 3 14 F. Interpretation of chemical shift of most elements – analysis of 3 factors: accessibility of excited states, distance of electron cloud & multiple bonding 1. Exceptions - anomalous upfield effects expected for any nucleus, other than 1H, with heavy atom substituents a) Methyl bromide 10 b) Methylene bromide 22 c) Bromoform 12 d) Carbon tetrabromide –29 2. I series monotonic in upfield shift with increased number of iodine atoms 3. Called "heavy atom effect" a) Attributed to spin-orbit coupling b) Nuclear spins shielded (upfield shift) by currents associated with electrons in orbits that are unsymmetrical & in motion IV. Carbon Chemical Shifts and Structure A. Saturated Alkanes 1. No pi bond - minimize p 2. effect - each additional methyl, add 9 PPM a) Cyclopropane -2.6 b) Methane -2.1 c) Ethane 5.9 d) n-propane 16.1 e) Isobutane 25.2 3. Beta effect C - Y - H -> C - Y – X 03/06/16 SCHM 530, Chapter 3 4. Gamma effect C - Z - Y - H -> C - Z - Y – X a) Important stereochemical component 5. Because of alpha, beta & gamma, alkane shift range large 6. = -2.5 + Aini a) –2.5 for methane b) Substituent effects up to 5 bonds away c) Complications (1) Corrections for branching d) Gamma effect is negative (1) Anti effect +1 (2) Gauche effect –6 (3) Average value –2.5 B. Cyclic Alkanes 1. Cyclopropane –2.6 2. Cyclobutane 23.3 3. Remaining cycloalkanes within 2 PPM of cyclohexane 27.7 4. Formula differ due to chair conformations C. Functionalized Saturated Alkanes 1. Inductive effect of radial term – shifts to higher frequency (downfield) a) Strong electron withdrawing group - large alpha effect b) CH3X (1) F 75.4 15 03/06/16 SCHM 530, Chapter 3 (2) Cl 25.1 (3) Br 10.2 (4) I -20.5 c) CH3OH 49.2 -CH2OH 49-75 -CH2OR 59-75 d) Methylamine in aqueous solution 28.3 (1) Range for amines extending some 30 PPM to higher frequency (2) Dimethyl sulfide 19.5 (3) Acrylonitrile at 0.3 (4) Nitromethane at 57.3 16 03/06/16 SCHM 530, Chapter 3 2. Not strongly effected by C=C a) 17.3 for methyl in trans-2-butene b) Toluene 21.3 c) Range 10-40 for carbons on double bonds d) Range when attached to carbonyl 30-45 e) Alpha effect largely due to electronegativity 3. Table 3-5, p 52 D. Alkenes 1. 100-170 a) =CH2 ~100 b) =CHR ~120 c) =CR2 ~150 2. 2-cyclohexenone C2 at 128.4, C3 at 149.8 3. 2-cylopentenone C2 at 132.9, C3 at 164.2 4. Calculate based on shift of ethene – 123.3 E. Alkynes and Nitriles 1. #CH 67-70, 2. #CR 74-85, 3. -C#N 117-130 4. n --> * transition pushes range to high frequency F. Aromatics 1. Toluene a) C1 at 137.8 - ipso 17 03/06/16 SCHM 530, Chapter 3 18 b) C2 at 129.3 c) C3 at 128.5 d) C4 at 125.3 2. Nitrobenzene – conjugation of nitro to ring a) C1 at 148.3 b) C2 at 123.4 c) C3 at 129.5 d) C4 at 134.7 e) ipso C - no H, longer relaxation time, lower intensity G. carbonyl groups 1. 160-220 2. Lower intensity 3. Aldehydes 190-205 4. Ketones 195-220 5. Carboxylic acid derivatives155-185 6. Esters 165-175 7. Acids 170-185 8. Acid chlorides 160-170 9. Anhydrides 165-175 10. Amides 160-175 11. Oximes 145-165 12. Central atom for allenes 200-215 V. Summary and tables of chemical shifts 03/06/16 SCHM 530, Chapter 3 19 A. Solvent effect 1. Saturated hydrocarbons – van der Waals forces a) Minimal effect 2. Polar (acetone, chloroform, diethyl ether, DMSO, acetonitrile) & nonpolar (carbon tetrachloride) interact through electrostatic interactions 3. Solvents with large diamagnetic anisotropy a) Separate overlapping resonances b) Benzene, toluene, nitrobenzene, nitroethane (1) Flat shape c) Acetonitrile, carbon disulfide, sulfur dioxide (1) Rod like, opposite effect 4. Examples a) HC#CCH2Cl the CH2 is at (1) 3.87 in cyclohexane (2) 4.17 in acetonitrile (3) 3.42 in benzene b) the Hs at C1 & C2 in pyrole are (1) Overlapping in benzene (2) Well separated in cyclohexane, acetone