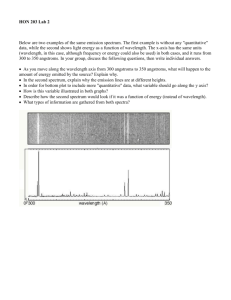
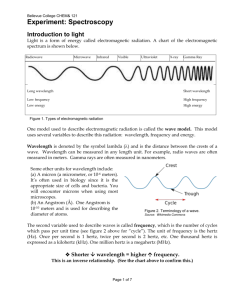
The purpose of this lab is: to understand emission spectra
advertisement

Chemistry: Spectroscope Lab - The Electromagnetic Spectrum Purpose: The purpose of this lab is: to understand emission spectra; to discover the different spectra that are created from different light sources (containing different elements); to practice converting angstroms into meters for wavelengths and to practice calculating energy associated with a specific photon of light. The numbers on the spectroscope scale represent 1,000 Angstroms (Ex. 5.20 = 5200 angstroms). 1.0x 1010 angstroms =1 meter. Observe each light source listed. Record your observations being as specific as possible. (Start with bright line or continuous) 1. a. Observe the continuous spectrum emitted from a white fluorescent light. b. List the order of the color bands from left to right, make sure you have the spectroscope held correctly. c. What is the wavelength of the orange band in the fluorescent bulb in Angstroms? Convert the wavelength to meters. Calculate the frequency of the orange photon. Calculate the energy associated with the orange photon. 2. Observe a 2 different colored light bulbs. How do they compare with each other? How are they the same or different than the white fluorescent bulb? 3. Observe the spectrum of the Hg vapor light source (it’s above you!). a) Describe what you see. Is it a bright-line spectrum or continuous spectrum? b) Calculate the wavelength of the green band in meters. c) Calculate the frequency of the green photon. d) Calculate the energy of the green photon. 4. Observe and record your observations of each of the following gases using your spectroscope. Use the chart provided to record which color bands are visible and the wavelength of each. a. Hydrogen:__________________________________________________________ b. Neon:____________________________________________________________ c. Helium:________________________________________________________________ d. Hydrogen shows 3 color bands and yet it only has 1 electron. Explain how this can be. e. How are the lines of the bright line spectrum being created? Analysis and Conclusion Questions: 5. Explain how the diffraction film (or a prism) on the spectroscope produces colors. 6. Which has the larger wavelength, a photon of red or violet light? Which of these two has the highest frequency? 7. What relationship (equation) did Planck find relating energy and frequency of a wave? 8. Which of the two colors in the above equation has the greater energy? 9. The line spectrum and the flame test are unique (different) for each element. Why? 10. Now that you know about the gas found in fluorescent tubes, why is there so much concern about proper disposal of these bulbs? 11. How do scientists use spectroscopy? 12. Helium was discovered by spectroscopy studies of the sun. Why do you think that it was so hard to detect helium on earth before this method came along? (This is a thought question, just make a guess. What do you know about helium?)