Isolation and determination of optically pure carvone enantiomers
advertisement

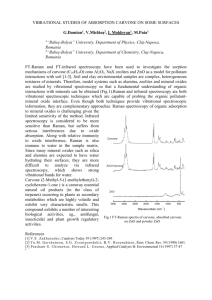
FLAVOUR AND FRAGRANCE JOURNAL, VOL. 2, 95-97 (1987) Isolation and Determination of Optically Pure Carvone Enantiomers from Caraway (Carum carvi L.), Dill (Anethum graveolens L.), Spearmint (Mentha spicata L.) and Mentha longifolia (L.) Huds. Uzi Ravid, Michal Bassat and Eli Putievsky Department of Medicinal. Spice and Aromatic Plants, Agricultural Research Organization. Newe Ya'ar. Haifa Post 31-999, Israel Vera Weinstein and Raphael Ikan Department of Organic Chemistry, Natural Products Laboratory, Hebrew University of Jerusalem, Jerusalem 91-904, Israel Optically pure (S)-( +)-carvone was isolated from the essential oils of the fruits of caraway and dill. Optically pure (R)-(-)-carvone was isolated from the essential oils of the leaves of spearmint and Menfha longifolia. Determination of the enantiomeric purity of the natural flavour was performed by 'H-NMR spectroscopy using a chiral lanthanide shift reagent, Eu(hfc)3. KEY WORDS (S)-( +)-Carvone (R)-(-)-Carvone Carum carvi L. Anelhum graveolens L. Mentha spicata L. Mentha longifolia (L.) Huds. 'H-NMR Chiral Eu(hfc)3. INTRODUCTION EXPERIMENTAL [(2-methylJ-(l-methylethenyl)-2-cycloCarvone hexene-1-one] occurs in nature in the (S)( + ) , ( R ) - ( - 1 and (RS) forms. (R)-(-)-Carvone is a major component of spearmint and kuromoji oils and is synthesized commercially from ( R ) (+)-limonene. It has been found in Menrha spicata, M . viridis var. crispa, M . longifolia, Eucalyptus globulus and several other mint species. .' (S)-(+)-carvone is a major component in caraway and dill oils. It has been also detected in Anethum sowa, Lippia carviodora, Mentha arvensis and others.'.* It has been found that ( R ) - ( - ) carvone, which has a spearmint odour, is detectable at a lower threshold than its enantiomer, which has a caraway odour.%' The (RS) form is present in gingergrass, Listea guatemafeusis, lavender and Artemisia ferganensis.'.* (S)-(+)Carvone is the starting material for the synthesis of (R ,Z)-3-met hy l-6-isopropen yl-3,Pdecadien- 1yl acetate, a pheromone component of the female California red scale.' (R)-(-)-Carvone is used as a starting material in the preparation of picrotoxinin.' Caraway (Carurn carvi L.), dill (Anethum graveolens L.), spearmint (Mentha spicata L.) and M. longifolia (L.) Huds. were grown in an experimental field at the Newe Ya'ar Agricultural Experiment Station. Seeds of commercial varieties of caraway and dill received from Holland, were sown in the autumn of 1984. Mentha longifolia was collected in 1984 from a garden and transferred to the Newe Ya'ar experimental field. A voucher specimen is retained at Newe Ya'ar. A commercial variety of M . spicara was received from a nursery in Rumania in the summer of 1980. The fresh foliage of the Mentha species was steam-distilled for 1 h in a 130-L direct steam pilot plant apparatus. Ripe fruits of caraway and dill were handseparated, ground, and hydrodistilled in a modified Clevenger-type apparatus for 2 h. The isolated oils (yields: caraway 3.6%, dill 3.8%, M. spicata 1.0%, M . longifolia 0.7%) were dried over anhydrous sodium sulphate and stored at 44°C.Carvone was isolated from the oils by flash chromatography under pressure ( 2 atm.) on silica gel Davisil 60 A, 200-425 mesh (Aldrich Chemic- ' 0882-5734/87/030095-03$05 .OO 0 1987 by John Wiley & Sons, Ltd. Received 15 May 1987 96 U . R A V I D , M. R A S S A T . E. PUTIEVSKY, V . WEINSTEIN A N D R . IKAN al Co.), eluant: 1 to 5% solution of ethyl acetate in hexane: and by fractional distillation in a Perkin-Elmer model 131 T microstill at 0.2-0.4 mbar. The chemical purities of natural and commercial samples of carvone were detected by a Varian 3700 gas chromatograph equipped with + FID, and a Hewlett-Packard 3390A integrator. Samples of 0.1 pl of the natural fractions and of commercial carvone were injected on a packed glass column (3m x 4mm i.d.) with 5% Carbowax 20M on acid-washed, silanized Chromosorb W (80-100 mesh) and on a fused silica capillary column (25 m) coated with Carbowax 20M. Operating conditions were: temperature programme, 7&200"C (5°C min-I); carrier gas, nitrogen, 30 and S m l min-', respectively. A commercial sample of (S)-( +)-carvone was obtained from Carl Roth and Co. ([a]:' + 54.0"; . c = 5.25, EtOH) and (R)-(-)-cawone was PI. 8 7 6 5 4 obtained from R. C. Treatt and Co. ([a];' -59.1'; c = 3.50. EtOH). Tris (3- Fig. I . ' H - N M R spectra of (S)-(+)-carvonc [LI/[S] = 0.7-0.8 (heptafluoropropylhydroxymethy1ene)-( )camphorate], europium ( I l l ) [ E u ( h f ~ ) ~ ]was , obtained from Aldrich Chemical Co. Optical rotations were measured on a Perkin-Elmer Model 531 polarimeter. 'H-NMR spectra were obtained at 200MHz with a Bruker Model WP-2OOSY Fourier transform spectrometer. Carvone (lOpl) w a s dissolved in a solution of 2% TMS in deuterochloroform (350 pl) in the NMR tube. Ten-milligram aliquots of Eu(hfc)? were added and dissolved in the solution. Molar ratios of the lanthanide shift reagent to substrate ([L]/[S]) were 0.45, 0.60, 0.75, 0.90, 1.05 and 1.20. . L ...... -. 2 3 with Eu(hfc)l iit + RESULTS AND DISCUSSION Carvone is the major constituent in the essential oils of the ripe fruits from caraway (51.2%) and dill (59.3%). and of the fresh leaves from two Metirliu species (69.9% in M. spicata and 61.6% in M. longifoliu). 'H-NMR spectra of the purified natural ( S ) - (+)-carvone isolated from caraway and dill. and of the (R)-(-)-cawone isolated from M. .spicuru and M. lotigifoliu in the presence of Eu(hfc), at increasing lanthanide-to-substrate molar ratios ([L]/[S] = 0.45 t o 1.20), show that there is n o separation of enantiomeric proton signals, so the carvone from the four oils is enantiomerically pure (Figures 1.2). The singlet of the the allylic C-7 methyl protons (1.70 ppm in v1.8 7 6 . . 5 . . 4 . . . 3 Fig. 2. ' H - N M R spectra of (R)-(-)-cawone with Eu(hfc), at [ L ] I [ S ]= 0.7-0.8 the unshifted spectrum) shows a significant downfield signal at 6.02 ppm for the (S)-(+)carvone and at 5.97 ppm for the (R)-(-)-cawone, at [L]/[S] = 1.20. The singlet of the allylic C-10 methyl protons moves downfield to 2.24 ppm at the same [L]/(S] ratio. The large downfield changes in the chemical shifts of the two methyl OPTlCALLY PURE CARVONE ENANTlOMERS An I I . ~ 7 6 5 i, 4 97 phenyl-2-pentanone, using europium chiral shift reagents of [L]/[S] = 1.O-2.0. were in the range of 0.03 to 0.75."."' Natural oxygenated monoterpenes isolated from essential oils are enantiomerically ure in the case of (-)-linalol from sweet basil' and (-)-linalyl acetate from clary sa e, Salvia dominica, lavender and lavandin.82 (+)-Linalol from coriander' I and (+)-terpinen-4-01 from sweet marjoramI3 are mixtures of the two enantiomers with the (+)-enantiomer predominant. (R)-(-)-Carvone is synthesized commercially from readily available (R)-(+)-limonene by nitrosochlorination. dehydrochlorination and hydrolysis" with retention of the integrity of the chiral centre. Routine detection of the enantiomeric purity of this important nature identical flavour by 'H-NMR using Eu(hfc)3 is a simple, fast and accurate technique. 3 Fig. 3. 'H-NMR spectra of (RS)-carvone with Eu(hfc), at [L]/[S] = 0.7-0.8 protons are due to the fact that the C-7 methyl is much closer to the binding site of the ketone (C=O) to the europium chelating agent than is the C-10 methyl. The (S)-(+)-carvone was spiked with the (R)-(-)-enantiomer in a ratio of 1:1, in the presence of Eu(hfc)3 (Figure 3) at [L]/[S] = 0.45 to 1.20. The absorption of the C-7 methyl protons is split into two equal signals at [L]/[S] = 0.45 (AAd = 0.01) to [L]/[S] = 1.20 ( A A b = 0.05). There is no splitting of the absorption of the C-10 methyl protons at the same lanthanideto-substrate molar ratios. Spiking of the two enantiomers in a 1:2 ratio shows that the absorption of the C-7 methyl protons at the higher field is that of the (R)-(-)-enantiomer. Spiking eliminates errors due to changes in the [L]/[S] ratio, the temperature, and other factors controlling the downfield position of the singlet by showin both enantiomers with one enhanced.' 'H-NMR spectra of commercial samples of ( S ) - ( + ) - and (R)-(-)-cawone, in the presence of Eu(hfc)z at molar ratios of 0.45 to 1.20, show that the samples were enantiomerically pure. The magnitudes of the enaiitiomeric shift differences of the methyl protons of the ketones camphor, 3-methyl-2-pentanone and 3-methyl-3- Acknorc~/e~~ernieirr-Contribution from the Agricultural Research organization. The Volcani Center, Bet Dagan. Israel. No. 2052-E. 1987 series. REFERENCES I . The Merrk Index, ed. M. Windholz, p. 239. Mcrck Ct Co.. N . J . (1976). 2. T. E. Furia and N. Bellanca (eds.). Feiiuroli's Huiidhook of Fluvour Ingrrdierirs. 2nd edn. Vol. 2. p. 87. CRC Press. Cleveland, Ohio (1975). 3. L. Friedman and J . G . Miller, Scietice. 172. I044 (1971). 4. G. F. Russel and J . I. Hills, Science. 172, 1043 (1971). 5. T. J. Leitereg. D. G. Guadagni, J. Harris. T. R. Mon and R. Teranishi. Nulure, 230. 455 (1971). 6. W. Roelofs, M. Gieselmann, A. CdrdC, H. Tashiro, D . S. Moreno. C. A. Henrick and R. J . Anderson. J. Chem. Ecol., 4. 211 (1978). 7. E. J . Corey and H. L. Pearce. 1. A m . Cbern. Soc.. 101. 5841 (1979). 8. E. L. Plurnmer, T. E . Stewart. K. Byrne. G . T. Pearce and R. M. Silverstein. 1.Cbetn. Ecol., 2, 307 (1976). 9. M. D. McCreary, D. W. Lewis. D. L. Wernick and G. M. Whitesides. 1.A m . Chern. Sor., %. 1038 (1974). 10. H. L. Goering. J . N . Eikenberry. G. S. Koermer. and C. J . L a t h e r . J. Am. Cherti. Soc., 96, 1493 (1974). 1 1 . U . Ravid. E. Putievsky. V . Weinstein and R. Ikan i n Essenriul Oils mid Aroniuric Plritirr, ed. A . Baerheirn Svendsen and J . J. C. Scheffer. pp. 135-138, Martinus NijhofUDr. W. Junk. Dordrecht (1985). 12. U. Ravid, E. Putievsky, M. Bassar. R. Ikan and V. Weinstein, Flav. frugr. j., 1, 121 (1986). 13. U . Ravid, E. Putievsky. M. Bassat. R. lkan and V. Weinstein, Flav. frugr. j . (accepted). 14. B. Singararn and J. Verghese. Perfurn. Fla r... 2,17 (1977).