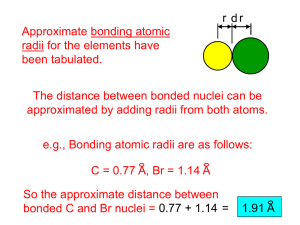
Atomic Radius –Trend within a group
advertisement

Atomic Radius –Trend within a group Hydrogen (1s1) Atoms get larger going down a group Lithium (1s2 2s1) Sodium (1s2 2s2 2p6 3s1) Shielding Effect WHAT CAUSES THE ATOMIC RADIUS TO INCREASE AS YOU MOVE DOWN A GROUP? H Li As we increase the atomic number (or go down a group). . . each atom has another energy level, so the atoms get bigger. Na K Rb Atomic Radius-Trend within a period 1s2 2s2 2p1 1s2 2s2 2p2 5p+ Atoms get smaller as you move left-to-right across a period. 6p+ 1s2 2s2 2p3 7p+ Shrinking Effect What causes the atomic radius to decrease as you move across a period left-to-right? Going from left to right across a period, the size gets smaller. Electrons are in the same energy level. But, there is more nuclear charge. Outermost electrons are pulled closer. Na Mg Al Si P S Cl Ar Atomic Radii of Representative Elements (nm) 1A 2A 3A 4A Li Be B C 5A 6A 7A O F N Na Mg Al Si P S Cl K Ca Ga Ge As Se Br Rb Sr In Sn Sb Te I Cs Ba Tl Pb Bi Po At Atomic Radius Atomic Radius decreases H Li F Na K At Fr Decreases going up CHALLENGE 1. a. b. c. d. e. Arrange the following groups of atoms in order of decreasing atomic radius: B, Al, Ga Sn, Sb, Te Cd, Si, Ga As, P, Cl O, Cl, F ANSWERS 1. a. Ga, Al, B b. Sn, Sb, Te c. Cd, Ga, Si d. As, P, Cl e. Cl, O, F WHAT ARE IONS? Atoms can gain or lose electrons. When they do, they form ions. An ion is an atom (or group of atoms) that has a positive or negative charge. IONS Metals tend to LOSE electrons, from their outer energy level. Removal of the electron form positive ions (atoms with a “+” charge) are called “cations.” (pronounced “cat-eye-on”) Nonmetals tend to GAIN one or more electrons. Adding electrons form negative ions (atoms with a “-” charge) are called “anions.” (pronounced “an-eye-on”) IONS Sodium loses one: there are now more protons (11) than electrons (10). The charge is written as a number followed by a plus sign: Na1+ Now named a “sodium ion” Chlorine will gain one electron Protons (17) no longer equals the electrons (18), so a charge of -1 Cl1- is re-named a “chloride ion” WRITE THE SYMBOLS AND CHARGES FOR THE FOLLOWING ELEMENTS: Element Calcium Bromine Lithium Chemical Symbol Charge WRITE THE SYMBOLS AND CHARGES FOR THE FOLLOWING ELEMENTS: Element Chemical Symbol Charge Calcium Ca Ca2+ Bromine Br Br1- Lithium Li Li1+ Ionization Energy Energy needed to remove one of atom’s electrons from its outermost shell A+E A+ + e- H Li F Na K At Fr Ionization energy increases Ionization energy increases Ionization Energy H Li F Na K At Fr Electronegativity increases Ionization energy increases Electron Affinity increases Electron affinity increases Ionization energy increases Atomic Radius increases/ Ionic size Electronegativity increases Summary of Periodic Trends