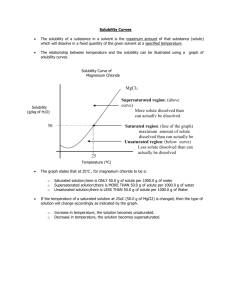
Solubility Curves
advertisement

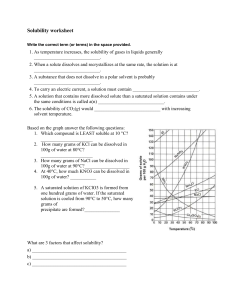
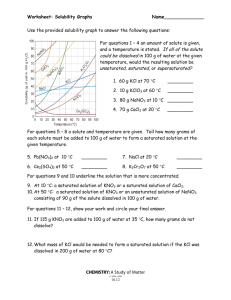
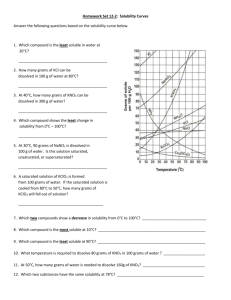
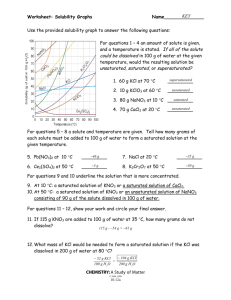
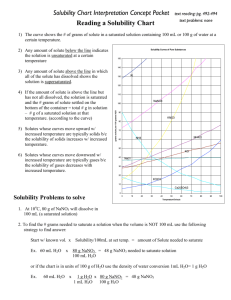
Solubility Curves 1. Which substance does not appear to increase greatly in solubility as the temperature is increased? 2. Which substance appears to decrease in solubility as the temperature is increased? Reading a solubility chart 1. 2. 3. 4. 3. Which substance is the most soluble at 0C ? 4. Which substance is the least soluble at 0C ? 5. 5. Which two substances have the same solubility at 71C ? 6. 6. What is the most KNO3 that can be dissolved in 100 ml of water at 70C ? 7. The curve shows the # of grams of solute in a saturated solution containing 100 mL or 100 g of water at a certain temperature. Any amount of solute below the line indicates the solution is unsaturated at a certain temperature. Any amount of solute above the line in which all of the solute has dissolved shows the solution is supersaturated. If the amount of solute is above the line, but has not all dissolved, the solution is saturated and the # grams of solute settled on the bottom of the container = total # grams in solution - # g of a saturated solution at that temperature. (according to the curve) Solutes whose curves move upward with increasing temperatures are typically solids because the solubility of a solid increases with increased temperature. Solutes whose curves move downwards with increased temperature are typically gases because the solubility of gases decreases with increased temperature. Use a proportion when the water given is larger or smaller than 100 mL or 100 g 7. What is the most NH3 that can be dissolved in 100 ml of water at 70C ? 8. At 70C which substance, KNO3 or NH3, can form the more concentrated solution? 9. At what temperature would you need 100 ml of water to dissolve 100g NaNO3 ? 10. If 100ml of a saturated solution of NH4Cl at 100C is cooled to 20C, how much precipitate will form? 11. If 100 ml of a saturated solution of KNO3 at 50C is cooled to 10C, how much precipitate will form? 12. 200 ml of a solution contains 10g of KClO3 at 15C. The solution is then warmed to 100C. How much additional KClO3 may be dissolved? 13. How many grams of NaNO3 are required to saturate 200ml of water at 10ºC? 14. A 100ml solution contains 35g KClO3 and 40g of NH4Cl at 80C. As the solution is cooled, which will precipitate first? 15. Classify the following solutions as either: saturated, unsaturated or supersaturated (assume that all three could form supersaturated solutions) a) 40. g of KCl in 100 mL of water at 80oC b) 120. g of KNO3 in 100 mL of water at 60oC c) 80. g of NaNO3 in 100 mL of water at 10oC SOLUBILITY CURVES FOR A NUMBER OF WATER SOLUBLE INORGANIC SUBSTANCES