#16 How does atomic size change within groups and across periods?
advertisement
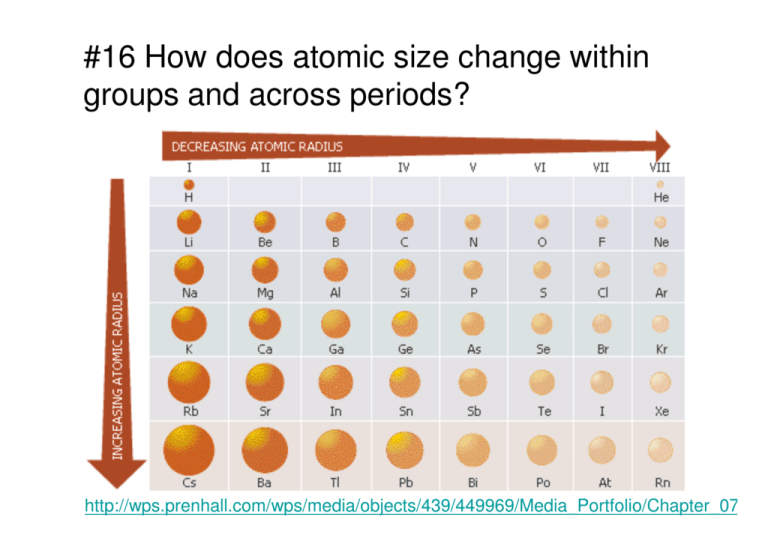
#16 How does atomic size change within groups and across periods? http://wps.prenhall.com/wps/media/objects/439/449969/Media_Portfolio/Chapter_07/Per #17 When do ions form? #18 What happens to first ionization energy within groups and across periods. Within groups it tends to be similar and decreasing. Across periods it tends to increase dramatically #19 Compare the size of ions to the size of the atoms from which they form. #20 How does electronegativity vary within groups and across periods. http://wps.prenhall.com/wps/media/objects/439/449969/Media_Portfolio/Chapter_07/PerT #21 In general, how can the periodic trends displayed by elements be explained? Differences in electron configuration http://center.acs.org/periodic/tools/PT.html #22 Arrange these elements in order of decreasing atomic size: sulfur, chlorine, aluminum, and sodium. Does your arrangement demonstrate a periodic trend or a group trend? Na Al S Cl a Periodic trend #23 Which element in each pair has the larger first ionization energy? a. sodium or potassium? Na b. magnesium or phosphorus? P











