“Anaerobic Respiration Lab” Introduction Ethanol is a fuel produced
advertisement

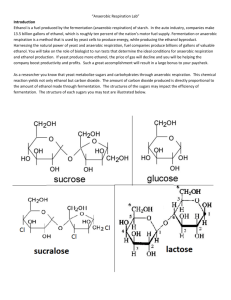
“Anaerobic Respiration Lab” Introduction Ethanol is a fuel produced by the fermentation (anaerobic respiration) of starch. In the auto industry, companies make 13.5 billion gallons of ethanol, which is roughly ten percent of the nations motor fuel supply. Fermentation or anaerobic respiration is a method that is used by yeast cells to produce energy, while producing the ethanol byproduct. Harnessing the natural power of yeast and anaerobic respiration, fuel companies produce billions of gallons of valuable ethanol. You will take on the role of biologist to run tests that determine the ideal conditions for anaerobic respiration and ethanol production. If yeast produce more ethanol, the price of gas will decline and you will be helping the company boost productivity and profits. Such a great accomplishment will result in a large bonus to your paycheck. Prelab Questions 1. Compare and contrast anaerobic and aerobic cellular respiration. 2. Note the equation of anaerobic respiration. 3. How does anaerobic respiration differ in our muscles and yeast? Experimental Question: How do different carbohydrates such as table sugar (sucrose) and glucose affect the rate of ethanol production by the anaerobic respiration of yeast? Hypothesis: Procedure 1. In a dosing syringe, obtain exactly 5 mL of the glucose yeast solution. 2. Attach an empty dosing syringe with a tube to the syringe containing yeast solution. 3. Squeeze about half of the solution into the other syringe. 4. Repeat steps 1-3 with table sugar (sucrose). 5. Wait and record the total volume of Carbon Dioxide produced by anaerobic respiration every two minutes. 7. Continue to take measurements over two minute intervals for the remainder of class. Data: Create a table, and graph to represent your data. An example of a table and graph has been provided below. Time (minutes) “Respiration Rate of Yeast” Volume of Carbon Dioxide Volume of Carbon Dioxide (sucrose) (glucose) 3 6 9 12 15 18 Etc. “Yeast Carbon Dioxide Production from Anaerobic Respiration” 7 6 Volume of Carbon Dioxide (mL) 5 4 Sucrose 3 Glucose 2 1 0 2 4 6 8 Time (minutes) Conclusions 1. Make your recommendations on what sugar should be used to maximize the production of ethanol. Refer to your data to support your conclusions. 2. Summarize the process of anaerobic respiration. 3. Predict any sources of human or experimental error that may have affected your results.