Synthesis of Dilantin Introduction: Dilantin is commonly used to treat
advertisement

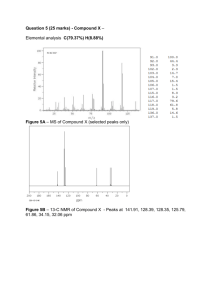
Synthesis of Dilantin Introduction: Dilantin is commonly used to treat epilepsy. It is an enzyme inducing drug that helps metabolize other anticonvulsant drugs in the body which slows impulses in the brain that causes seizures.1 Dilantin reacts with many other drugs because it has some very unique properties. It is loosely bounded to plasma proteins, and largely metabolized by cytochrome enzymes. Since it is loosely bound to these enzymes and proteins, the occurrence of inhibition and metabolism with other drugs is frequent. The main mechanism of dilantin is inhibiting collagenase. This is very useful when treating skin related diseases and injuries. A specific example of the effect of dilantin on a cellular level is examining patients with epidermolysis bullosa. People who suffer from this disease have an increased level of collagenase which removes polysaccharides from the connective tissue and increases blistering.2 By inhibiting collagenase with dilantin, blistering of the skin decreases. Additionally, dilantin is known to increase the expression of growth factor B in macrophages which promotes the healing of ulcers and traumatic wounds.2 Dilantin has many different uses and can treat a variety of diseases which makes it a very essential drug in the medical field. Dilantin is synthesized by a condensation reaction between urea and benzil which is also known as the Bilitz synthesis. This experiment uses a modified version of the traditional condensation reactions because the yields are not as high as they should be. The first step in the reaction is addition of the urea anion to benzil which rearranges in a fashion similar to a benzilbenzilic rearrangement.3 Next, the oxygen group becomes protonated by water and leaves behind a base in solution. This base is protonated by the proton of the secondary amine and water is reformed. Electron shifts occur and the hydroxyl group is lost. This hydroxyl group deprotonates the primary amine and causes cyclization. Finally, the benzylic rearrangement occurs which completes the synthesis of Dilantin. Dilantin can be synthesized by a condensation reaction which is a technique that is readily used in organic chemistry. A condensation reaction is when 2 or more molecules combine to form a larger structure with a loss of a smaller molecule such as water. There are many types of condensation reactions: aldol, claisen, carbonyl, dieckmann, and even polymerization. Many important drugs and materials are synthesized by this reaction which makes it an essential technique in organic chemistry. The purpose of this experiment is to synthesize dilantin from benzil and urea via a condensation reaction. The crude product will then be purified by recrystallization. The success of the experiment will be determined by calculating a percent yield, determining the melting point, and analyzing the final product by IR, 60 MHz 1H NMR, 400 1H NMR, and 400 MHz 13C NMR. Experimental: Benzil (400 mg), absolute ethanol (6 mL), 30% aqueous sodium hydroxide (1.5 mL), and urea (200 mg) was refluxed for 2 hours. This reaction was monitored every 30 minutes by TLC. Once completed, the mixture was cooled and poured into distilled water (10 mL). It was then acidified drop wise using glacial acetic acid. Once acidity was determined by pH paper, it was placed on ice (15 mins) and collected via vacuum filtration. The product was then purified by recrystallization (95% ethanol); 1H NMR (400 MHz, DMSO) δ (ppm) 7.3471-7.4035 (m, 10H), 9.3397(s, 1H), 11 (m, 1H); 13C NMR (400 MHz, DMSO) δ (ppm) 38.6913-39.9435, 126.4583, 127.9442, 128.4149, 155.9083; IR (ATR) v max (cm-1) 1491.88, 1679.06, 1770.43, 2108.06. Results and Discussion: The synthesis of dilantin was done by a condensation reaction. Urea and Benzil were combined together to for a denser compound. The high pressure in the condenser allows the starting materials to come together and form the desired product. This reaction was done in an alkaline solvent, and the presence of sodium hydroxide allowed a phenyl shift to occur. This reaction was monitored by TLC. Dilantin is much more polar that benzil and therefore eluted faster in the nonpolar mobile phase. Once there was no evidence of starting material, the reaction was stopped and acidified to protonate the alkene to yield the final product. From the 0.4 grams of benzil, 0.505 grams of Dilantin is theoretically supposed to be produced. Before recrystallization, 0.3950 grams of Dilantin was collected. However, after the purification some product was lost. 0.3028 grams was the final weight of the recovered product which makes the percent recover 77% and the percent yield 60%. This exceeded the percent yield in a typical traditional synthesis (55%), but is not as high as it should be in the Bilitz synthesis. This may have been from product being melted during the process of recrystallization. The melting point of the final product was also determined to analyze the success of the reaction. Dilantin has a high melting point of 293-295°C, and benzil has a significantly lower melting point of 94-95°C. The observed melting point was higher than 150 degrees Celsius but could not be determined directly because it surpassed the thermometers maximum temperature reading. However, since the product did not melt at 94-95°C, it can be concluded that benzil was not present. This product was analyzed by IR spectra. The major change that should have been observed was the transformation of a carbonyl group into cyclopentanone and the addition of amines. The amine was a weak signal at 1491.88 cm-1, and the cyclopentanone was a strong sharp signal at 1770.43 cm-1. Since there was no presence of a peak at 1715 cm-1, this indicated that carbonyl groups were not present from the starting material benzil. This spectrum also shows the presence of the benzene ring double bonds at 1679.06 cm-1, and the benzene hydrogens at 3208.06 cm-1. The presence of these particular peaks does characterize dilantin and therefore can be concluded that the reaction did proceed as planned. 1 H NMR 60 MHz was not very helpful in characterizing the product. Therefore the product was analyzed by 1H NMR 400 MHz because it is more sensitive and can show the peaks more clearly. There was a large water impurity at 3.443 ppm that may have been in the tube prior to adding the sample, and a peak at around 2.5 which represents the DMSO solvent and can be ignored. The multiple peaks around the 7 ppm zone represent the 10 aryl hydrogens. This does not differ from bezil, but can still be used to confirm Dilantin is present. Additionally, the peaks at 9.3397 ppm and 11 ppm represent the 2 amide protons. No amide protons are present in benzil so this shows that the reaction did go to completion. 13 C NMR was also useful in characterizing the final product. There is a peak at 155.9083 ppm which represents the carbonyl group, peaks around 127 ppm which represent the phenyl carbons, and a peak at 70.1062 ppm which represents the tetrasubstituted carbons. There are 7 distinct carbon peaks the 39 ppm range. This indicates that Dilantin is in fact present and not benzil because benzil only has 5 distinct carbons. Overall, the data from this experiment showed that the desired product was synthesized by a successful condensation reaction. The IR, H NMR, and C NMR data characterized dilantin, and the melting point and percent yield further supported the synthesis of the product. There were a few minor impurities that could be fixed by being more careful while preparing the NMR samples. References: 1. J. Chem. Educ., 1983, 60 (6), p 512 2. The Journal of Investigative Dermatology (1951) 16, 43–52; doi:10.1038/jid.1951.6 3. J. Chem. Educ., 1986, 63 (7), p 650