The Michaelis – Menten Equation kcat/Km
advertisement

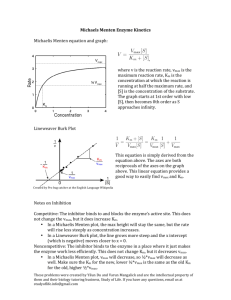
Previous Class •Michaelis – Menten equation •Steady state vs pre-steady state Today Review derivation and interpretation ¾Graphical representation ¾Michaelis – Menten equations and parameters The Michaelis – Menten Equation v = Vmax [S] Km + [S] Km = Michaelis constant: Concentration of Substrate needed to reach half maximum velocity – measure of substrate affinity Vmax = maximum velocity – directly proportional to enzyme concentration The Michaelis – Menten Equation Km Ef + S k1 k-1 ES k2 Ef + P [E][S] = k-1 + k2 = Km [ES] k Eqn 1 1 Km is an apparent dissociation constant (Ks) and represents the [S] when v = ½ Vmax Therefore, a lower Km value indicates a higher affinity for the substrate The Michaelis – Menten Equation The Michaelis – Menten Equation Interpretation Obtain kinetic behaviour of an enzyme E+S k2 = kcat kcat = Vmax/[ET] k1 k-1 ES k2 E+ P Catalytic constant of the reaction (first order) when k2 is fast (saturating kinetics) (When EP → E + P is fast) kcat is also known as the turnover number of the enzyme – defining the maximum number of substrate molecules converted to product per unit of time Units v0 = initial velocity of Product formation = moles Product formation (moles substrate loss)/litre x time = mM s-1 Vmax = represents the maximum rate the enzyme reaction can achieve. Vmax occurs when all of the enzyme is in the ES complex. Km = [S] at Vmax/2 = µM kcat = first order rate constant = s-1 # of catalytic cycles active site undergoes per unit of time The Michaelis – Menten Equation kcat/Km At any [S] including: At very low [S] ([S]→0) : pre-steady state conditions v0 = Vmax [S] Km + [S] becomes v0 = Vmax[S] = kcat[E][S] Km Km The second order rate constant kcat/Km indicates the catalytic efficiency of the enzyme: A direct measure of the efficiency of the enzyme in transforming Subst. kcat/Km combines: the effectiveness of transformation of bound product the effectiveness of productive substrate binding Units v0 = initial velocity of Product formation = moles Product formation (moles substrate loss)/litre x time = mM s-1 Vmax = represents the maximum rate the enzyme reaction can achieve. Vmax occurs when all of the enzyme is in the ES complex. Km = [S] at Vmax/2 = µM kcat = first order rate constant = s-1 # of catalytic cycles active site undergoes per unit of time kcat/Km = second order rate reaction of E and S = M-1 s-1 The Michaelis – Menten Equation Lineweaver-Burke transformation of the Michaelis-Menton equation. •Velocity vs substrate plots are useful for visually estimating kinetic parameters •Hyperbolic curves cannot properly determine the upper limit of the curve (Vmax) •Transforming the data to a form that can be plotted as a line. Lineweaver-Burke transformation of the Michaelis-Menton equation. v = Vmax [S] Km + [S] Reciprocal of the equation is: 1 Km + [S] = v Vmax [S] Express reciprocal in the familiar form y = mx + b Lineweaver-Burke transformation of the Michaelis-Menton equation. y = mx + b 1 V = Km Vmax [S] + 1 Vmax 1 V Km Vmax [S] = + 1 Vmax Lineweaver-Burke equation represents a straight line with slope = Km/Vmax, y intercept = 1/Vmax, and x intercept = -1/Km A 0.6 B 0.5 1/v (mM/min) 0.4 0.3 0.2 0.1 0.3 0.25 0.20 0.15 0.10 0.1 1/[S] (mM) 0.15 0.2 0.25 0.30 0.35 Lineweaver-Burke transformation of the Michaelis-Menton equation. •Most commonly used •Magnitude of errors can become distorted •Furthest point to the right (lowest [S]) influences where line is drawn •e.g. small error in v = large error in 1/v •Good for observing enzyme inhibition Eadie-Hofstee Plot •Multiply both sides of L.B. by Vmax •Multiply both sides by v •Rearrange for v v0 v K m 0 Vmax = [S] Plot v0 vs v0 [S] Eadie-Hofstee Plot Vmax v0 -Km = slope Vmax Km v0/[S] Eadie-Hofstee Plot •Best for controlling slight deviations from linearity •One disadvantage is that the least precise parameter (v0) is expressed in both sides of the equation and plot Plotting kinetic data Michaelis-Menten kinetics (steady state kinetics) Measure Initial rates (v0) at different substrate concentrations by detecting the absorbance difference over time and obtaining the slope of the line Obtain various initial rates at different substrate concentrations and plot Use Lineweaver-Burke plots to obtain kcat, Km, kcat/Km Plotting kinetic data Measuring initial rates: •The initial velocity is the amount of product produced per minute •Assay involves adding all of the components including substrate first •Abs vs time can be plotted •Determine slope of tangent Absor. •Once enzyme is added the absorbance is continually monitored and recorded with respect to time Enzyme added time Plotting kinetic data Measuring initial rates: •The slope of tangent = ∆Abs/unit of time (min) •Recall A = εcl, therefore ∆A = ε∆cl (change in conc of Product) •When ∆c occurs in a known time period then ∆c min-1 = v0 •v0 = ∆A min-1/ εl = ∆c min-1 •Has M/min after multiplying by vol. of enzyme assay solution the units change to mol/min. Plotting kinetic data Determine v0 at different substrate concentrations and plot