Self Assessment: Physical and Chemical Properties of Elements
advertisement

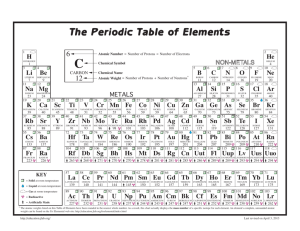
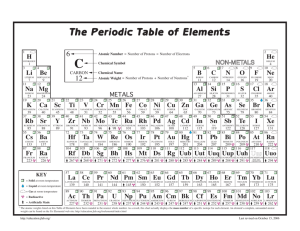
Name: ____________________________________ Class: _____________ Date: _____________ Self Assessment: Physical and Chemical Properties of Elements 1. Which of the following statements is true? a) Elements within a group tend to have similar chemical properties. b) Elements within a period tend to have similar chemical properties. c) Elements of a similar boiling point tend to have similar chemical properties. d) Elements of a similar melting point tend to have similar chemical properties. 2. An unknown element is a gas at room temperature. It is not known to react with any other elements. What group does it likely belong to? a) Group 18 b) Group 17 c) Group 1 d) Group 2 3. A group of scientists studies the properties of four elements. The scientists find that all four elements are soft, shiny metals that are very reactive. Also, they all have relatively low densities. They differ significantly in their atomic masses. What conclusion could the scientists draw about these elements? a) They all belong to the same group. b) They all belong to the same period. c) They all have the same atomic number. d) They all melt at the same temperature. 4. Iodine is located four places to the right of indium in Period 5. Given this information, which statement is most likely to be true? a) Iodine has a larger atomic radius than indium. b) Iodine has a lower ionization energy than indium. c) Iodine has a lower atomic number than indium. d) Iodine has a greater electronegativity than indium. PAGE 1 Name: ____________________________________ Class: _____________ Date: _____________ Self Assessment: Physical and Chemical Properties of Elements 5. How might an element in the upper right portion of the periodic table differ from an element in the lower left portion of the periodic table? a) The element in the upper right would have a larger atomic radius. b) The element in the upper right would be more reactive. c) The element in the upper right would have a greater electronegativity. d) The element in the upper right would have a greater atomic number. 6. Atoms of chlorine, bromine, and fluorine each have seven valence electrons, while an atom of magnesium has only two valence electrons. What property is magnesium not likely to share with chlorine, bromine, and fluorine because of this difference? a) ability to form ionic compounds b) reactivity with elements from Group 1 c) ability to gain or lose electrons in a reaction d) inability to react with elements from Group 18 7. Which group of properties describes most metals? a) dull in appearance, poor conductor, solid at room temperature b) shiny in appearance, good conductor, solid at room temperature c) shiny in appearance, good conductor, gas at room temperature d) dull in appearance, good conductor, solid at room temperature 8. Imagine that element X is in Group 2 of the periodic table. What compound is element X likely to form with halogen A? a) XA2 b) XA c) X2 A3 d) X2 A 9. How are elements arranged in the periodic table? a) by increasing atomic number b) by increasing atomic mass c) by increasing atomic radius d) by increasing atomic weight PAGE 2 Name: ____________________________________ Class: _____________ Self Assessment: Physical and Chemical Properties of Elements 10. Which of the following describes a physical property of elements in Group 2? a) react with halogens in 2:1 ratios b) react with noble gases in 2:1 ratios c) gas at room temperature d) shiny color PAGE 3 Date: _____________ ANSWER KEY 1. a 2. a 3. a 4. d 5. c 6. b ANSWER KEY Page 1 7. b 8. a 9. a 10. d