formula of a precipitate experiment
advertisement

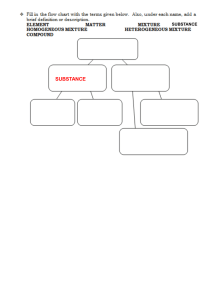
name: _______________ date: _______ determine the ratio of cations and anions in a compound - experiment introduction: Atoms of different elements will lose and gain electrons and stick together to form ionic compounds, also known as ‘salts.’ The formula for an ionic compound provides the ration of cations to anions; for example, in NaCl, there is a 1:1 ratio of sodium to chlorine while in CaF2, there is a 1:2 ratio of calcium to fluorine. In this experiment, the ratio of cations and anions for the compound formed between copper (II) ions (Cu2+) and phosphate ions (PO43-) will be experimentally determined. materials and equipment: safety goggles, test tube rack, test tubes (7), ruler, copper (II) chloride solution, sodium phosphate solution, beaker(s), Sharpie marker(s), plastic pipet(s) procedure: 1. Label 7 small test tubes (#1 - #7) using a Sharpie marker and place them in a test tube rack. 2. Obtain a small beaker with about 6 mL of the copper (II) chloride solution and a small beaker with about 6 mL of the sodium phosphate solution. 3. Carefully add the appropriate number of drops of each solution to test tubes 1 - 7 as indicated by the table below. It is extremely important that the correct number of drops go into each test tube. If you lose count, wash out the test tube and start over. test tube 1 2 3 4 5 6 7 drops of copper (II) chloride 3 6 12 15 18 24 27 drops of sodium phosphate 27 24 18 15 12 6 3 ratio of CuCl2 to Na3PO4 4. Cap each test tube and carefully shake each mixture. Let the test tubes sit undisturbed in the rack for at least 10 minutes for the precipitate to settle to the bottom. During this time, calculate the ratio of drops of copper (II) chloride to sodium phosphate and fill in the bottom row of the table above. 5. After all of the precipitate has settled (at least 10 minutes), observe both the solid at the bottom and the liquid above it and record your observations in the table below. Be as detailed as possible. layer in test tube observations precipitate (solid) liquid above precipitate 6. Using a ruler, measure the height (in millimeters) of the precipitate and record it in the data table below. 7. Dispose of the contents of each test tube in a sink, rinse each test tube, clean up all materials, and wash your hands once the experiment is completed. data and results: test tube 1 2 3 4 5 6 7 height of precipitate (mm) 1. Write the formula for the neutral ionic compound formed between copper (II) ions and phosphate ions. 2. Predict the ratio of copper (II) ions to phosphate ions, based on the formula you wrote above. 3. Create a bar graph with ‘test tube number’ on the x-axis and ‘height of precipitate in mm’ on the y-axis. 4. Which test tube had the greatest amount of precipitate? What was the ratio of drops in that test tube? Does that ratio match your prediction in question 2 above? 5. Did any test tubes show evidence of unreacted copper (II) ions in the liquid above the precipitate? Hint: Did the liquid appear to have a blue tint? 6. What was the total number of drops of solution in each test tube? Why was it necessary to keep the total number of drops constant?