Levi's Copy of Formula of an Ionic Compound Lab Honors - Google Docs
advertisement

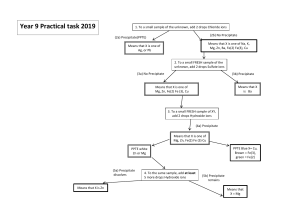
Introduction Title: Formula of an Ionic Compound Purpose Statement: The purpose of this experiment is to determine the empirical formula of an unknown ionic compound. :Hypothesis: If we mixed equal amounts of reactant ions in a reaction, then the amount of precipitate formed can be plotted against the volume ratio and as a result the empirical formula can be determined. Procedure: Two solutions containing equal amounts of two reactant ions will be combined in a series of reactions. In each reaction, the total volume of the two solutions will be held constant while the volume ratio of the reactant is varied. The amount of precipitate obtained in each reaction will be measured and plotted against the volume ratio to find the empirical formula of the product. Variables Independent Variable: The volume ratios of the reactants Dependent Variable: Volume (amount) of the precipitate Control Variable:The accepted empirical formula of the precipitate Constants (5): Total volume of the two solutions, the concentration of the solutions, Pre-lab Assignment: 1. Many common drugstore chemicals are ionic compounds. Use the internet to write the IUPAC name correct empirical formula for each of the following compounds: a. milk of magnesia: magnesium hydroxide: Mg(OH)2 b. washing soda : sodium carbonate: Na2CO3 c. epsom salt magnesium sulfate: MgSO4 2. Solution of iron (III) chloride and sodium hydroxide were mixed in a series of precipitation reactions as described in this experiment: a. Name the two possible products in this precipitation reaction and predict their empirical formulas. iron (III) chloride: FeCl3 sodium hydroxide: NaOH iron (III) chloride + sodium hydroxide → Iron (III) hydroxide + sodium chloride FeCl3(aq) + NaOH (aq) → Fe(OH)3(ppt) + NaCl(aq) b. Which product is likely to be insoluble in water and precipitate out as a red solid? The iron (III) hydroxide is likely to be insoluble in water and forms the red solid because we know that NaCl is table salt and salt dissolves in water. c. What volume ratio of reactants gave the most precipitate? Explain Test tube # Volume ratio( drops of Cu to drops of Na) ==Height of precipitate (mm) 1 2 1:9 1:4 0.20 3 2:3 0.76 0.72 4 5 1:1 3:2 1.35 1.28 6 7 4:1 9:1 0.64 0.98 Data Table: Initial and final observations of the reaction Color of FeCl3 Solution light Honey Color of NaOH solution Appearance of Product orangey brown clear Data Table: Precipitation Reactions Test Tube # Volume ratio (drops of Fe to drops of Oh) 1 1:11 2 1:5 3 1:4 4 1:3 5 6 7 Height of precipitate (mm) 0.56 1.01 0.98 1.46 1.42 1.56 1.42 Post-lab Questions: Answer the following questions in complete sentences and thought. Show your work including all significant figures and units in each answer. 1. Use the data from the lab to answer the following questions: a. Give the formula and names of the two possible products in the precipitation reaction of copper (II) chloride with sodium phosphate. Copper Phosphate (Cu4(PO4)2) and Sodium Chloride ( NaCl) b. Based on common knowledge, which product is likely to be insoluble in water and to precipitate from the solution. (Use the solubility rules if you need to) The Copper(II) Phosphate is likely to be insoluble because sodium chloride dissolves in water. 2. Use EXCEL to create a graph to show the height of the precipitate in each test tube. 3. Which test tube had the greatest amount of precipitate. Does this result agree with the prediction made in Question #1 concerning the empirical formula of the product? Explain. The test tube with the greatest amount of precipitate was test tube #5, with 14mm of precipitate. It had a 2:3 Copper (II) Chloride solution to Sodium Phosphate solution ratio, which demonstrates that when the solution contained higher levels of Copper (II) Chloride, there was a greater amount of precipitate. 4. Write a balanced chemical equation for the precipitation reaction of copper (II) chloride and sodium phosphate. Include abbreviations for the physical state of each reactant and product, using (aq) for aqueous solution, (s) for solid, (l) for liquid, and (g) for gas. 3CuCl2(aq) + 2Na3PO4(aq) à Cu3(PO4)2 (s) + 6NaCl(aq) 5. a. Which test showed evidence of unreacted Cu+2 ions in the supernatant when the reaction was complete? Explain why unreacted Cu+2 ions were present in these tubes based on the volume ratio of solutions used. Test tubes #6-72-6 contained a blue supernatant, with evidence of the Cu2+ ions present. b. How could you tell that all of the Cu+2 ions had reacted in a particular test tube? Which test tube showed such evidence. Explain, based on the volume ratio of solutions used. Test tubes 1-5 We could tell that the Cu2+ ions had reacted been present because of the formation of a clear supernatent. 6. What was the total number of drops of solution in each test tube? Why was it necessary to keep the total volume of reactants constant in each test tube? Ideally, there would be exactly 30 drops of solution in each tube. However, due to experimental error, some of the drops were not equal sizes. It was important to keep the total volume of the reactants constant because then the reactions could be observed equally. 7. Does the height of the precipitate in each test-tube accurately reflect the amount of precipitate in each case? Hint: Compare the shape of the test tube with that of a graduated cylinder. What effect does this error have on the conclusions reached in this experiment. The height of precipitate does not accurately reflect the amount of precipitate in each case because the test tubes do not have a flat bottom and thus are not uniform in shape. This would mean that the ruler measured more precipitate than there truly was. 8. Suggest one improvement that would increase the accuracy and precision of results for this lab. Be specific in your answer.