Temperature, Heat and Internal Energy
(A) Temperature and thermometers
Temperature is a measure of the hotness of a body.
(1) Temperature scale and thermometer
Temperature is expressed using a temperature scale. A temperature scale is
obtained by choosing two fixed points, and dividing the range between them into a
number of equal divisions called degrees.
The most commonly used temperature scale is the Celsius temperature scale.
Thermometer is a device for measuring temperature. Each type of thermometer
uses a physical property that changes with temperature. For example, in a
liquid-in-glass thermometer, the liquid expands and rises up in the narrow tube
when temperature rises.
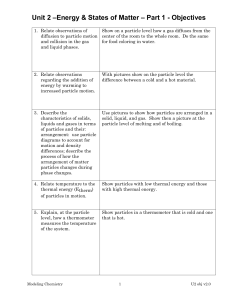
(2) Temperature and Particle Motion
According to the kinetic theory, all matter is made up of very tiny particles, which
are constantly in motion. When they are close together, they attract/repel each
other strongly. When they are far apart, they hardly attract/repel each other at all.
Kinetic theory can be used to explain the general properties of solids, liquids and
gases.
Solids
Liquids
Gases
General property
Arrangement of particle
Fixed volume and Particles
are
close
shape
together are arranged in
fixed positions
Fixed volume but Particles are also close
no fixed shape
together, but they are not
in fixed positions.
Movement of particles
Each
particle
only
vibrates to and fro about
a fixed position.
Each particle can move
from one place to
another.
No fixed volume Particles are very far Each particle can move
and shape
apart. There is almost no from one place to
attractive force between another.
them.
The average kinetic energy of particles in an object increases with temperature of
the object. Two bodies have the same temperature if particles in each body have
the same average kinetic energy.
Temperature, Heat and Internal Energy
1
(B) Heat and internal energy
When two bodies of different temperatures touch each other, energy is transferred
from the hot body to the cold body until they reach the same temperature. (The
bodies are then said to be in thermal equilibrium.)
Heat is the energy transferred from one body to another as a result of a
temperature difference.
Unit of heat: joule (J)
Internal energy is the energy stored in a body. It increases when the temperature
of the body rises, or when the body changes from solid to liquid or from liquid to
gas.
Internal energy is the sum of kinetic and potential energy of all particles in the
body.
Unit of internal energy: joule (J)
Power means the rate of energy transfer, i.e.
Power
Energy
Time
Unit of power: watt (W) or joule per second (J s-1)
(C) Heat capacity and specific heat capacity
(1) Heat capacity
Heat capacity (C) is the energy transferred to raise the temperature of the object
through 1 °C.
Heat capacity = mass × specific heat capacity
or
C = mc
Unit of heat capacity: J °C–1
(2) Specific heat capacity
Specific heat capacity (c) is the energy transferred to raise the temperature of 1 kg
of the substance through 1 °C.
Energy transferred = mass × specific heat capacity × change in temperature
or
E = mcT
Unit of specific heat capacity: J kg–1 °C–1
Temperature, Heat and Internal Energy
2
(3) Measuring specific heat capacity
Measuring specific heat capacity of Measuring specific heat capacity of
water
aluminium
Experimental
set-up
c
Calculation
E
mT
c
E
mT
Do not switch on the heater Add a few drops of oil to the holes in
unless its heating part is totally
the aluminium block.
immersed in water.
Precaution
Keep the heating part of the
Place the aluminium block on a
heater totally immersed in water
polystyrene tile.
throughout the experiment.
Do not take the final temperature
as soon as the power supply is Do not switch on the heater unless
its heating part is in contact with the
switched off. Stir the water and
aluminium block.
record the highest reading.
Possible
sources
error
of
Some energy is lost to the Some energy
surroundings.
surroundings.
is
lost
to
the
Some energy transferred is used
to heat up the polystyrene cup,
the stirrer and the thermometer.
(4) Importance of high specific heat capacity of water
Water is a good choice for use as a coolant.
Coastal areas have cooler summers and milder winters than inland areas of
the same latitude.
The body temperature only changes slowly when the temperature of the
surroundings changes.
Temperature, Heat and Internal Energy
3
 0
0