Beer Lambert`s law, Raman spectroscopy
advertisement
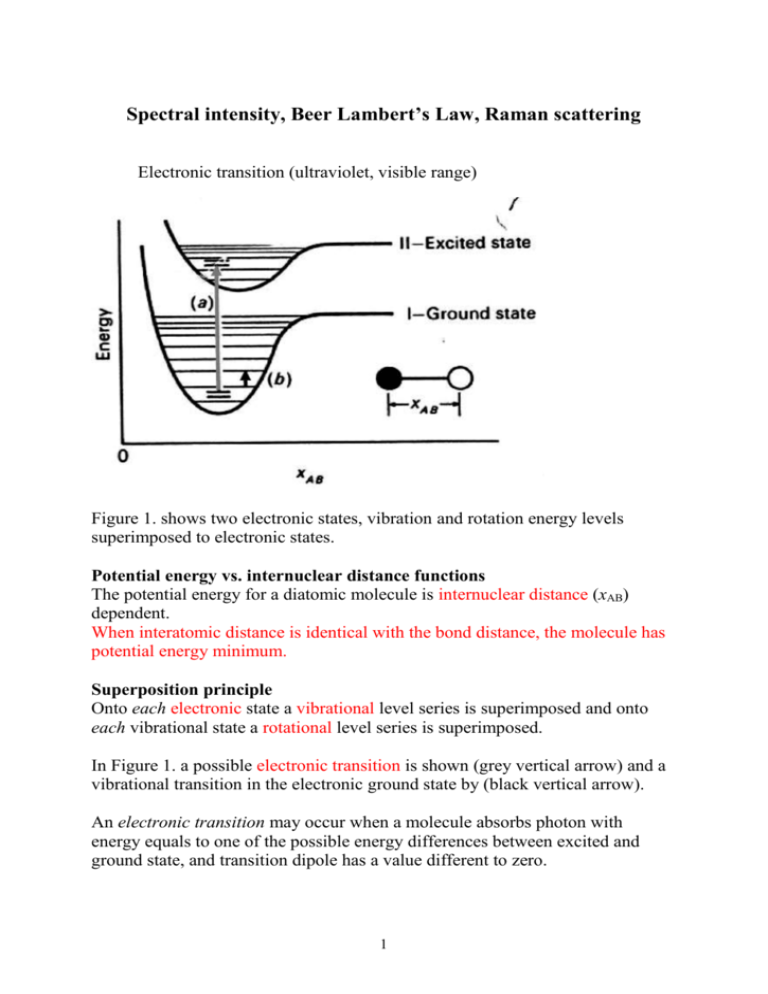
Spectral intensity, Beer Lambert’s Law, Raman scattering Electronic transition (ultraviolet, visible range) Figure 1. shows two electronic states, vibration and rotation energy levels superimposed to electronic states. Potential energy vs. internuclear distance functions The potential energy for a diatomic molecule is internuclear distance (xAB) dependent. When interatomic distance is identical with the bond distance, the molecule has potential energy minimum. Superposition principle Onto each electronic state a vibrational level series is superimposed and onto each vibrational state a rotational level series is superimposed. In Figure 1. a possible electronic transition is shown (grey vertical arrow) and a vibrational transition in the electronic ground state by (black vertical arrow). An electronic transition may occur when a molecule absorbs photon with energy equals to one of the possible energy differences between excited and ground state, and transition dipole has a value different to zero. 1 Absorption of light, Beer-Lambert’s law The light intensity functions The flux of photons, Φ is defined as the number of photons, np transmitted through a cross sectional area, q during time, t. np q t If the source is monochromatic the equation can be transformed to energy flux multiplying the number of photons by hν which gives photon intensity: I h h np q t . 1. 1a. unit: Wm-2. The incoming radiation intensity I0 decreases when transferring an optically active sample. The outgoing radiation intensity, I equals the difference in I = I0 – I a 2. between incoming and absorbed intensities, accordig to the law of energy conservation Figure 2. The radiating power or intensity is reduced by an absorbing medium of b optical path. In photometry, we use relative intensities, dividing Eq. 1. by I0 we get I Ia 1 I0 I0 3. 2 a quantity called transmittance. I T I0 The percentage transmission, T% 4. T % 100 T Beer-Lambert law Increasing the thickness l (optical pathlength) of an absorbing layer by dl the transmitted intensity decreases by dI. Therefore, this derivative dI dl is negative, and directly proportional a material coefficient ε, the outgoing intensity, I and the concentration of light absorbing material c in the solution,. dI I c dl 5. Integrating this equation between limits l 1 d I c I 0 I 0 dl I the integrated function ln I ln I 0 c l Rearranging and introducing the wavelength dependence ln I0 A c l I The function form: A c l 6. This form assign variables (asorbance, molar absorbance) dependence of other variables. Be careful is not a product! The absorbance at constant wavelength is linearly dependent on the concentration of absorbing material, optical path and specific absorbance (ε (λ)). 3 When concentration is given in mol dm-3 the coefficient ε is called molar absorbance. The usual non SI unit of thickness is cm, and the unit of ε is dm3mol-1cm-1. ε depends on the nature of material, the matrix and the wavelength of radiating source. (Matrix refers to the components of a sample other than the analyte of interest.) Intensity formulas When the material is transparent: I = I0, T = 1 or T% = 100. When the material absorbs all the incoming light: I = 0 or T% = 0 The logarithmic ratio of intensities called absorbance is frequently applied in quantitative determinations. I 7. A log10 0 I The output data of spectrophotometers are transmittance, T transmittance percentage, T% or absorbance, A. Absorbance can be given in terms of transmittance, 1 A log10 log10 T T 7a. or in terms of transmittance percentage A log10 100 log10 100 log10 T % 2 log10 T % T% From intensity functions only the absorbance is directly proportional to the concentration of analyte, therefore it is frequently used for concentration determination. The light absorbing components determine the absorbance of a multicomponent system. E.g. dissolving iodine in CCl4 a violet coloured solution is obtained for which the absorbance at 520 nm (the maximum of iodine absorption) is: CCl solution iodine A520 520 cCCl l 520 ciodine l 4 4 At 520 nm the CCl4 is transparent thus 520 0 CCl 4 4 solution iodine A520 520 ciodine l At constant c and l a spectral band can be observed. By increasing concentration a band series is obtained. 0,5 Concentration series b = const. = const. Absorbance 0,4 0,3 0,2 0,1 0,0 200 300 400 500 600 Wavelength / nm Figure 3. concentration series of an absorption band centered at 520 nm. Concentration dependence 0,5 Absorbance 0,4 max = 400 nm b = const. 3 = 833,3 dm /(mol cm) 0,3 0,2 0,1 0,0001 0,0002 0,0003 0,0004 0,0005 0,0006 0,0007 3 Concentration / mol/dm Figure 4. Translation of Fig. 3 at a single wavelength. The linear concentration dependence 5 We read absorbance data at λmax from band series and plot the concentration dependence of absorbance. As BL law is valid in the concentration range studied, we have a straight line in Figure 4. As BL law contains no additive constant the ideal graph crosses the x axis at the origin i.e. at A=0, c=0 point. Experimental error causes the value of intercept to be different from zero. Example 1. The molar absorption coefficient of a substance dissolved in hexane is known to be 855 M-1cm-1 at λ = 270 nm. Calculate the percentage reduction in intensity when light of that wavelength passes through 2.5 mm of a solution of concentration 3.25 10-3 M. Solution Excess datum: λ = 270 nm l = 0.25 cm c = 0.00325 M ε = 855 M-1cm-1 Percentage reduction = T% A c l I A lg 0 , I T% 1 A 100 10 I0 10 A I T% . 100 10cl T% 100 20.2 106.947 Example 2. Riboflavine in dilute acetate solution shows a maximum in absorption spectrum at 444 nm. Calculate the wavenumber in cm-1 and frequency of light at this peak. Calculate the energy of corresponding electronic transition for one molecule and one mole of riboflavin. Data: h = 6.626 10-34 Js, c = 3.00 108 ms-1, NA = 6.022 1023. λ = 444 10-9 m = 444 10-7 cm ~ 1 22522.5 cm-1 7 444 10 c 3 108 6.756 1014 s-1 9 444 10 1 For one molecule E h 6.626 10 -34 6 6.756 1014 4.476 1019 J 19 For one mole Emolar E N A 4.476 10 6,02 1023 269455.2 J/mol Example 3 A 2 mm path-length cell is filled 0.01 M benzene. The wavelength is set to maximum wavelength of benzene, 256 nm. At this wavelength the transmitted intensity was 48% of incident intensity. Suppose the solvent absorbance can be neglected. What is the molar absorbance and absorbance of benzene at this wavelength? c = 0.01 M l = 0.2 cm excess datum: 256 nm I = 0.48·I0 I lg 0.48 0.3187 I0 A 0.3187 159.35 cl 0.01 0.2 A lg Acid-base equilibrium of weak acid indicators Ionisation (dissociation) equilibrium of a weak acid: HA ↔ H+ + AThe equilibrium constant at dT = 0 and dp = 0 is H A K 8. HA Both the undissociated weak acid, [HA], and its dissociated anion, [A-], absorbs light at different wavelength (see Fig. 5.). 7 absorption band of A form 0,9 0,8 absorption band of HA form 0,7 Abszorbance 0,6 pH = 4,0 pH = 8,5 0,5 0,4 pH = 6,5 0,3 0,2 0,1 0,0 -0,1 350 400 450 500 550 600 isobestic 650 700 750 / nm Figure 5. Dissolved in a pH=4 buffer the weak acid is undissociated, in a pH=8.5 buffer solution it is in pure ionized form. At medium pH = 6.5 both components are present with appreciable intensity. Equation 8. can be transformed by introducing dissociation degree, α [c] = α·c0 The α fraction of the total indicator concentration (c0) is dissociated. The limits for α are: 0 1 . α K H 1 α 9. Turning to common logarithm lg K lgH lg α 1 α Introducing pH 8 lg K lg H lg pK pH lg α 1 α α 1 α or 10. Setting the pH of weak acid solution by a buffer the fraction of dissociation can be adjusted. When α = 0.5, from Eq. 10., pK pH The additivity of BL law If interactions among light absorbing components of a solution can be neglected, the measured absorbance at λ = constant is a sum of component absorbances. Ai A1 A2 ... An ε1c1l ε2c2l ... εn cn l 11. Turning back to the dissociation equilibrium applying the maximum wavelength of A- (see Fig. 5.). The BL law for the two species AHA εHA c0 l AA εA c0 l - Applying additivity rule A εHA 1 α c0l εA α c0l - A AHA 1 α AA- α 12. and for α α A AHA and AA AHA 1 α 1 A A A AHA A AA AHA AA AHA Substitute for α and 1- α to Eq. 10 we get 9 13. pK pH lg A AHA AA- A 14. containing measured data: A, AHA, AA- and pH. From analysis of Eq. 12 pK and consequently K can be determined. 1.Applying pH = 8.5 buffer solution (see Fig. 5.), the weak acid is totally dissociated, α = 1, and from Eq. 12. A AA2.Applying pH = 4 buffer solution, the weak acid is totally undissociated, α = 0, and from Eq. 12. A AHA 3. For medium pH Eq. 12. can be applied as it is. By knowing the pH, measuring AA-, A and AHA the equilibrium constant can be determined. This part of lecture series is a task in practical semester. Raman scattering, Raman intensities History Raman scattering is named after Indian physicist C. V. Raman who discovered it in 1928, though predictions had been made of such an inelastic scattering of light as far back as 1922. The importance of this discovery was recognised even then, and for his observation of this effect Raman was awarded the 1930 Nobel Prize in Physics. This was and remains the shortest time from a discovery to awarding of the Prize. Rayleigh scattering Most light passing through a transparent substance undergoes Rayleigh scattering. This is an elastic effect, which means that the light does not gain or lose energy during the scattering. Therefore it stays at the same wavelength. 10 In Rayleigh scattering a photon interacts with a molecule, polarising the electron cloud and raising it to a “virtual” energy state. This is extremely short lived (on the order of 10-14 seconds) and the molecule soon drops back down to its ground state, releasing a photon. This can be released in any direction, resulting in scattering. In the sense of energy conservation, the energy of emitted photon is the same as the incident one. Fig. 1. Energy level diagram of scattering processes Raman effect Raman scattering is different in that it is inelastic. The light photons lose or gain energy during the scattering process, and therefore increase or decrease in wavelength respectively. In Raman scattering, the energies of the absorbed and emitted photons are different. The Raman effect comprises a very small fraction, about 1 in 107, of the incident photons. Stokes scattering If the molecule is promoted from a ground to a virtual state and then drops back down to a (higher energy) vibrational state then the scattered photon has less energy than the incident photon, and therefore a longer wavelength. This is called Stokes scattering. Generally only the lowest vibrational level is occupied to any extent and the transition v =1 ← v = 0 with rotational states superimposed gives the strongest Raman band. The Stokes Raman frequency is displaced to a lower frequency from the Rayleigh scattered photon by a frequency equal to the vibrational frequency. 11 anti-Stokes scattering If the molecule is in a vibrational state to begin with and after scattering is in its ground state then the scattered photon has more energy, and therefore a shorter wavelength. This is called anti-Stokes scattering. An anti-Stokes line can be observed when photon collides a molecule in excited state, and a higher energy photon is emitted. That lines are less intense because of the population density of excited vibrational state is smaller than that is for ground state. Frequency shifts Rayleigh line (most intensive): incident scattered Stokes shift vibration incident scattered anti-Stokes shift vibration scattered incident A Stokes and anti-Stokes line belonging to the same vibration mode are equally displaced from Rayleigh line. The intensity of Raman scattering is inversely proportional to the fourth power of wavelength (IRaman ≈ 1/λ4). Selection rule: Recalling the definition equation of polarizability (defined in an earlier lecture of this course) μinduced α E The polarizability, α, represents the ability of an applied electric field, E, to induce a dipole moment, μind in an atom or molecule. For example, large atoms such as xenon have a strong polarizability because their electron clouds — distant from the xenon nucleus — are relatively easy to distort with an applied electric field. Helium atoms, which are smaller and more compact, have a small polarizability. Polarizabilities for atoms are isotropic (that is, the same in all directions), whereas polarizabilities for molecules may vary with position about the molecule, depending on the molecule’s symmetry. 12 Fig. 2. Schematic representation of the anisotropic polarisability of a molecule At a molecule’s equilibrium nuclear geometry, the polarizability is some value, α0. At some distance, Δr, away from the molecule’s equilibrium geometry, the instantaneous polarization α is given by 1. the derivative 1a. represents the change in polarizability with change in position. If that derivative equals zero, the entire second term is zero and there will be no Raman scattering. Thus, we have as a gross selection rule: a molecular motion will be Ramanactive only if the motion occurs with a changing polarizability. The schematic representations of the general set up of the monochromator based dispersion Raman spectrometer is given in 13 Fig. 3. The experimental setup of a Laser Raman spectrometer. Signal detection is done perpendicular to the direction of monochromatic beam. Raman spectrum of CCl4 The position of peaks of Stokes and anti Stokes are symmetrical Fig. 4. Laser source: argon ion laser, producing laser radiation of 488 nm. 14 Further reading Group frequencies When light is scattered from a molecule or crystal, most photons are elastically scattered. The scattered photons have the same energy (frequency) and, therefore, wavelength, as the incident photons. However, a small fraction of light (approximately 1 in 107 photons) is scattered at optical frequencies different from, and usually lower than, the frequency of the incident photons. The process leading to this inelastic scatter is termed the Raman effect. Raman scattering can occur with a change in vibrational, rotational or electronic energy of a molecule. If the scattering is elastic, the process is called Rayleigh scattering. If it’s not elastic, the process is called Raman scattering. Raman scattering (or the Raman effect) was discovered in 1928 by V. C. Raman who won the Nobel prize for his work. If the substance being studied is illuminated by monochromatic light, for example from a laser, the spectrum of the scattered light consists of a strong line (the exciting line) of the same frequency as the incident illumination together with weaker lines on either side shifted from the strong line by frequencies ranging from a few to about 3500 cm-1. The lines of frequency less than the exciting lines are called Stokes lines, the others anti-Stokes lines Raman spectroscopy is very important practical tool for quickly identifying molecules and minerals. A Raman spectrometer was deployed on the Viking landers in 1972 and in 15 other missions. Raman spectroscopy also has important scientific applications in studying molecular structure. In this experiment we will study both kinds of applications 16









