Exercise n:
advertisement

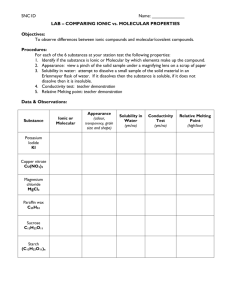
Name Lab section (day, AM/PM) 21 Exercise 5: Ionic and Molecular Compounds Introduction The type of bonding in a compound can greatly influence its chemical and physical properties. For example, ionic bonds allow such properties as solubility in water and electrical conductivity. Covalent compounds may or may not be soluble in water (depending upon their polarity) and many will burn or have a low melting point. The atoms and their three-dimensional arrangement (shape) in a molecular compound contribute to their solubility properties. Polar molecular compounds are usually soluble in water (like dissolves like), whereas non-polar molecular compounds are more likely to be soluble in a non-polar solvent like methylene chloride. Some molecular compounds will have both polar and non-polar portions and be soluble in both. Ionic compounds are more likely to have melting points than molecular compounds, and are solids at room temperature. In contrast, molecular compounds can be solid, liquid, or gas at room temperature, and even the solids are usually easily melted. The substances you will test are below: 1. Sodium chloride - NaCl 6. Ammonium nitrate - NH4NO3 2. Sucrose - C12H22O11 ( lots of -OH) 7. Toluene - C6H5CH3 3. Methyl alcohol - CH3OH 8. Isopropyl alcohol -CH3CHCH3 4. Benzoic acid - C6H5COOH 9. Potassium dichromate - K2Cr2O7 5. Sodium acetate - NaCH3COO 10. Copper(II) chloride - CuCl2 Procedures Part A. Descriptive 1. You will find jars of the solids being tested in the lab. In your Data Table write down the name, chemical formula, and two physical properties. Part B. Solubility Tests We often use the generalization “like dissolves like” to describe the solubility properties of polar and non-polar substances. Water is used as the polar substance here, and (CH2Cl2 ) methylene chloride as the non-polar solvent. 1. All test tubes must be clean and dry. For each substance to be tested, place 1 dropperful of methylene chloride (CH2Cl2) into a test tube. Add 2-3 drops (if a liquid) or several grains (if a solid) of the substance to the solvent. 2. Mix the contents and observe the results. DISCARD THE CONTENTS INTO THE WASTE CONTAINER - NOT THE SINK. Record the results in your Data Table. Name Lab section (day, AM/PM) 22 3. All test tubes must be clean free methylene chloride. For each substance to be tested, place 1 dropperful of distilled water into a test tube. Add 2-3 drops (if a liquid) or several grains (if a solid) of the substance to the solvent. 4. Mix the contents and observe the results. DO NOT DISCARD THE CONTENTS OF THE TEST TUBES. THESE SOLUTIONS WILL BE USED IN PART D. Record the results in your Data Table. Part C. Ignition Tests 1. Place about 1 mL of liquid, or a pea-sized portion of solid, in a clean, dry, COOL evaporating dish set on a wire gauze on a ring stand. 2. PLACE THE ENTIRE ASSEMBLY UNDER THE FUME HOOD. 3. Using the flame of a Bunsen burner, cautiously test each substance for melting, burning, or charring by placing the flame directly on the substance. (If a substance pops, but does not change physical state, it is probably just losing water. Record your observations in the Data Table. Part D. Conductivity 1. Check the results of your tests for solubility. For those substances that did not dissolve in water , place a negative sign in the Data Table under the column for conductivity, you will NOT test those for conductivity, assume no conductivity. 2. Rinse the electrodes of the conductivity meter. Using the conductivity meter placed near your work space, test the conductivity of the distilled water by placing 2 mL in a small test tube. (The distilled water should NOT conduct electricity, i.e., the light should not light. If it does, either the water is not as pure as we would like, the electrodes were not cleaned prior to use, or the test tube has the remains of a conducting substance in it. Clean everything and try again. If the distilled water still produces a weak light, even after you have cleaned everything again, note that at the bottom of the Report Sheet.) 3. For each substance that was soluble in water in part B (does not have a negative sign on the Report Sheet), dip the electrodes into the test tube and observe the conductivity meter. A bright light indicates that the solution containing the substance conducts electricity. Be sure to clean the conductivity probe between uses. Name Lab section (day, AM/PM) 23 Data Sheet for Exercise 5: Ionic and Molecular Compounds Solubility in Name and Formula Two Physical Properties CH2Cl2 Distilled Water Conductivity Test Results: H2 O Ignition Test Conductivity Name Lab section (day, AM/PM) QUESTIONS: 1. Of all of the tests and examinations of the compounds on the list, which test is the most reliable at discriminating between ionic and molecular compounds? 2. Which of the substances you tested were definitely ionic compounds? 3. Which of the substances you tested were definitely molecular compounds? 4. Were there any substances you tested that were difficult to decide? Why? 5. Summarize the physical characteristics of the various types compounds tested. IONIC POLAR COVALENT NON-POLAR COVALENT 24