Online microarray methods:
advertisement

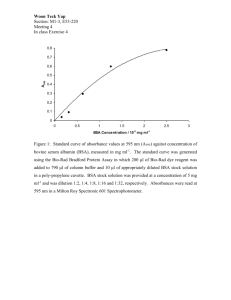
Online Appendix for the following April 1 JACC article TITLE: Hypoxia, Hypoxia-Inducible Transcription Factor, and Macrophages in Human Atherosclerotic Plaques Are Correlated With Intraplaque Angiogenesis. AUTHORS: Judith C. Sluimer, MSC, Jean-Marie Gasc, PHD, Job L. van Wanroij, MD, Natasja Kisters, BsC, Mathijs Groeneweg, MSC, Maarten D. Sollewijn Gelpke, MSC, Jack P. Cleutjens, PHD, Luc H. van den Akker, MD, Pierre Corvol, MD, PHD, Bradly G. Wouters, PHD, Mat J. Daemen, MD, PHD, Ann-Pascale J. Bijnens, PHD APPENDIX Methods THP-1 cell culture and differentiation. Human THP-1 cells were obtained from the American Type Culture Collection (ATCC 10801, Manassas, Virginia). Cells were grown in RPMI 1640 medium (Gibco-Invitrogen, Grand Island, New York) containing 10% FetalClone III (v/v; Hyclone, Pe-bio, Hogan, Utah), 1% GlutaMAX I (w/v; Gibco-Invitrogen), 125 mmol/l Hepes, penicillin (100 units/ml), and streptomycin (100 µg/ml; Gibco-Invitrogen) at 37°C in a humidified incubator with a 5% CO2 atmosphere. To differentiate THP-1 cells into macrophages, cells were seeded at a density of 1 106 cells/ml and incubated for 72 h with 0.2 μmol/l phorbol 12-myristate 13-acetate (Sigma, Saint Louis, Missouri). Microarray. Ribonucleic acid was isolated with the guanidine isothiocyanate/CsCl method (Chomczynski, 1987 #326). Ribonucleic acid quantity and quality were determined with a nanodrop spectophotometer (Witec AG, Littau, Switzerland) and a 2100 Bioanalyzer (Agilent Technologies, Palo Alto, California), respectively. All samples included had a RNA Integrity Number 5. Double-stranded cDNA was synthesized from 1.6 or 2.0 μg total RNA using the OneCycle Target Labeling Kit (Affymetrix, Santa Clara, California), and used as a template for the preparation of biotin-labeled cRNA with the GeneChip IVT Labeling Kit (Affymetrix). Biotinlabeled cRNA of 16 samples were hybridized to separate HGU133 2.0 Plus Arrays (Affymetrix), washed, stained with phycoerythrin-streptavidin conjugate (Molecular Probes, Eugene, Oregon), and the signals were amplified by staining the array with biotin-labeled anti-streptavidin antibody (Vector Laboratories, Burlingame, California) followed by phycoerythrin-streptavidin. The arrays were laser scanned with the GeneChip Scanner 3000 (Affymetrix) according to the manufacturer's instructions. Data were saved as a raw image file and quantified using GCOS 1.2 (Affymetrix). Pre-processing and normalization of signal intensities was conducted with the error model of Rosetta Resolver v5.1 (Rosetta Biosoftware, Seattle, Washington), which was developed for Affymetrix GeneChips. Technical and biological duplicates were combined to obtain intensities and standard errors per group. Rosetta Resolver and Locuslink (Entrez Gene) were used to combine the expression intensities of multiple probes per gene to obtain the expression intensity per gene. The error model of Rosetta Resolver was used to determine fold changes between early and advanced stable atherosclerotic lesions and were considered significantly different when p < 0.05. Ingenuity pathway analysis (Ingenuity Systems, Mountain View, California) was performed on genes with a p < 0.01. Online Table 1. Immunohistochemical Methods Antibody Company, Product Code Control Tissue Dilution Incubation Antigen Blocking Visualization Retrieval Pimonidazol NPI, Hypoxyprobe Normal skin 1 :50 1 h, RT ChemMate — Powervision-HRP HIF1a Becton Dickinson, #610958 CCRA 1:120 O/N, 4°C TE pH = 9 TBST/1%BSA Powervision-HRP Abcam, #463 CCRA 1:50 O/N, 4°C — TBST/1%BSA Powervision-HRP HIF2a Abcam, #8365 CCRA 1:750 O/N, 4°C ChemMate TBST/1%BSA Powervision-HRP VEGF Peprotech, #500-P10 ACA 1:50 O/N, 4°C — TBS/5%BSA Powervision-HRP GLUT1 Dako, #3536 COCA 1:100 1 h, RT ChemMate TBS/5%BSA Powervision-HRP GLUT3 Chemicon, #AB1345 Normal testis 1:500 1 h, RT ChemMate Biotin block Powervision-HRP HK1 Santa Cruz, #6517 Normal kidney 1:10 1 h, RT — TBST/1%BSA Powervision-HRP CD68 Dako, #M0814 ACA 1:100 30 min, RT Pepsin — Powervision-HRP CD31 Dako, #M0823 ACA 1:50 1 h, RT TE pH = 8 TBS/5%BSA Powervision-HRP/ABC-AP CD34 Monosan, #MON1164 ACA 1:200 1 h, RT TE pH = 8 TBS/5%BSA Powervision-HRP/ABC-AP Ki-67 Immunologic, #Rm9106-s ACA 1:25 1 h, RT TE pH = 8 TBS/5%BSA ABC-HRP Cleaved caspase 3 Cell Signaling, #9661 Tonsil 1:200 O/N, 4°C ChemMate TBS/5%BSA ABC-HRP ABC-AP/-HRP = alkaline phosphatase or horseradish-peroxidase conjugated avidin biotin complex (Vector laboratories); ACA = atherosclerotic carotid artery; BSA = bovine serum albumin; CCRA = clear cell renal carcinoma; ChemMate = ChemMate target retrieval; COCA = colon carcinoma; DAKO = Pepsin, 0.1% pepsin in 0.1N HCl; Powervision-HRP = horseradish-peroxidase conjugated Powervision goat anti-mouse antibody (Immunologic); TBST = Tris-buffered saline with 0.1% Tween; TE = Tris-EDTA buffer. Other abbreviations are explained in the text. Online Figure 1. No Pimonidazole Immunoreactivity in Control Patients (A) No pimonidazole immunoreactivity was detected in the carotid atherosclerotic plaques of a patient without pre-operative pimonidazole injection. Inset shows origin of magnification. (B) No pimonidazole immunoreactivity was detected in the macrophages of carotid atherosclerotic plaques of another patient without pre-operative pimonidazole treatment. Inset shows CD68 staining. (C) Hematoxylin and eosin staining of normal skin at incision site. (D) Hypoxia (pimonidazole immunoreactivity) is present in the epithelium, hair follicle, and sebaceous gland of normal skin tissue, a positive control for pimonidazole infusion.