Red Cabbage pH Indicator Lab: Test Acids & Bases
advertisement

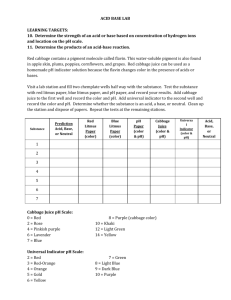
How to Make Red Cabbage pH Indicator and Test for Acids/Bases Acid-Base Chemistry Mad Science Lab Introduction: We know that acids and bases are chemicals that cause water to either lose hydrogen atoms (H+) or hydroxide atoms (OH-). The pH scale is used to measure whether a liquid is an acid or a base. Acids have low pH numbers and bases have high pH numbers. Acids and bases can be found almost anywhere, even in your own home! Question/Topic: Test some common household liquids to find out if they are acids or bases. We will make a pH indicator to determine whether a liquid is an acid or a base. An indicator is a chemical that changes color when added to acids or bases. Materials: Red Cabbage Distilled water White coffee filters 2 bowls Plastic cups Eye Dropper Some household liquids to test o Tap water o Vinegar o Lemon juice o Baking soda mixed with water o Soda (7-Up) o Seltzer Water o Ammonia o Soap and water o Other???? Experiment Procedure: Make the pH indicator: 1. Cut up the red cabbage into small pieces until you have about 2 cups 2. Put the cut up cabbage into one of the bowls and add boiling water. Let it sit for about 10 minutes 3. Strain the cabbage liquid through the coffee filter into the clean bowl. Throw away the used cabbage. Test for Acid or Base: 1. Mark each cup with the name of one of the test liquids from the lab table 2. Pour a SMALL amount of each test liquid into the marked plastic cups 3. In your lab books, make a hypothesis as to which liquids will be acids and which will be bases. 4. Using the eyedropper, slowly drip the cabbage indicator liquid into each cup. Observe the color of each liquid. Write down your observations in your lab book. 5. Write down the pH number that is related to the color Here is a table of approximate colors of red cabbage indicator solution at different pH's. pH Color 2 Red 4 Purple 6 Violet 8 Blue 10 Blue-Green 12 Greenish Yellow 6. Using the pH number, determine if each household liquid is an acid or a base. 7. Were your hypotheses valid? 8. Clean up lab area.