PCR Primer Design - Molecular Parasitology
advertisement

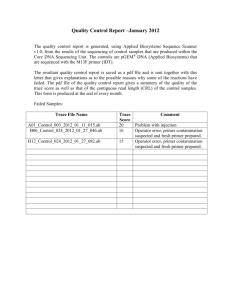
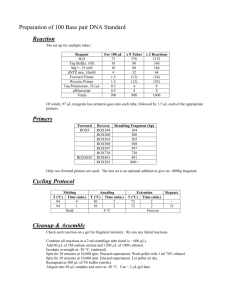
BIOL4905 Molecular Parasitology (Ulrich) Spring 2015 Primer Design Summary Instructions: In this assignment, you will report how you designed primers for your gene target and summarize primer characteristics and information relevant to amplification the gene by PCR. Follow the formatting guidelines and naming conventions exactly. 1. Selected Gene/Protein Target(s): List the names of the genes for which you are designing primers. (e.g. ORF_1234, ORF_2345, ORF_3456) 2. Brief (≤3 sentences) description of each protein: Describe in several sentences the characteristics of the protein you are studying (e.g. similarity to other proteins and potential function) 3. Name your primers with the following convention (“F” and R” are abbreviations for forward and reverse) i. CfFall14ORF_XXXX-Initials-F ii. CfFall14ORF_XXXX-Initials-R. 4. Use the following example (modified from Avraam Ardelean, a student from BIOL4905, Spring 2014) as a template for reporting information on one primer pair (one forward and matching primer) for each gene target. Please follow the formatting of this example. (Use your judgment on reporting relevant details on secondary structure and dimerization aspects of your primer, however. Remember that the example below is for informational purposes and the structure and dimerization information will vary from gene to gene.) ORF_4536 General Notes o NdeI and KpnI restriction sites were integrated for ligation into pNUS-GFPcH o The terminal stop codon of ORF_4536 was not included in the reverse primer sequence to allow fusion of ORF_4536 protein to GFP. Forward Primer CfSpring14ORF_4536-AA-F CATATGTGGAGGCGCTCTGTTCGCTG Tm = 65.2˚C o Restriction site (underlined): CATATG (NdeI) o Results of secondary structure prediction: One hairpin loop was predicted to occur between G8-C16. The energy required to denature this structure is -2.1 kcal/mole at a temperature of 45.4˚C, which is noticeably lower than the melting temperature (Tm) of the primer. o Notes on potential for homodimerization or heterodimer formation with the other primer: There are 11 potential homodimers that can form between two forward primers. The energy required to denature these homodimers ranges between -1.57 kcal/mole (2 bp hydrogen bond formations) and -9.89 kcal/mole (4 bp hydrogen bond formations). There are 18 possible heterodimer formations that can occur between the forward and reverse primers. The energy required to denature these heterodimers ranges between -1.34 kcal/mole (2 bp hydrogen bond formations) and -11.42 kcal/mole (5 bp hydrogen bond formations). Amplicon Size: 885 bp + 12 bp (restriction sites) = 897 Reverse Primer CfSpring14ORF_4536-AA-R GGTACCCTTCGCCTTGATCACCTCACCC Tm = 63.2˚C Restriction site (underlined): GGTACC (KpnI) Results of secondary structure prediction: Four prospective hairpin loops can occur within the reverse primer. They occur between the following base pairs in order of increasing energy required to denature the loop: 1. A11-T17 with a 2-bp helix stacked between G8-C18: 0.07 kcal/mole at 24˚C, 2. A11-T17 with a 3-bp helix stacked between T9-A19 and G10-C18: 0.28 kcal/mole at 20.3˚C, 3. C4-G10: 0.75 kcal/mole at 2.1˚C, and 4. C4-G10 with a 2-bp helix stacked between T3-A11: 0.99 kcal/mole at -4.9˚C. 5. All of these temperatures fall well under the Tm of the primer. Notes on potential for homodimerization or heterodimer formation with the other primer: There are five potential homodimers that can form between two reverse primers. The energy required to denature these homodimers ranges between -1.57 kcal/mole and -8.53 kcal/mole. The potential for heterodimerization between reverse and forward primers is akin to heterodimerization between forward and reverse primers, mentioned above. 5. Description of primer design methods used including, but not limited to tools and tool settings (e.g. length and Tm constraints): This is a short methods paragraph that reports how you designed the primers. 6. Cite any literature or software that you used. (If you used primer3, for instance, provide the citation for the software. You can typically find the appropriate citations on each tool’s website.) BIOL4905 Primer Design Summary Bioinformatics and Cell Biology Name: Date: Selected Gene/Protein Target(s): Brief (≤3 sentences) description of each protein: Primer Information (follow example provided in instructions) Description of primer design methods used: Literature Cited Fall 2014