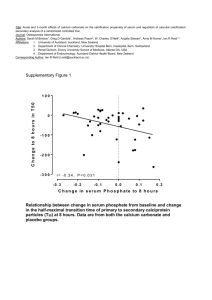
Introduction: Arterial vascular calcification
advertisement

P218 THE SELECTIVE P2X7 RECEPTOR ATAGONIST A438079 AMELIORATES VASCULAR SMOOTH MUSCLE CELL CALCIFICATION IN VITRO Booth, J, Norman, J, Wheeler, D, Unwin, R Centre for Nephrology, UCL Medical School, London INTRODUCTION: Arterial vascular calcification (VC) is highly prevalent in the advanced chronic kidney disease (CKD) population and is associated with increased mortality. VC is now recognised to result from an active process in the vessel wall: vascular smooth muscle cells (VSMCs) can adopt an osteoblastic phenotype in situ and mineralisation is controlled by the prevailing balance of calcification promoters (including a high calcium and phosphate milieu, inflammatory and oxidant stress, and cellular apoptosis) and inhibitory factors (such as pyrophosphate, matrix GLA protein and fetuin-A). The P2X7 purinoceptor (P2X7R) is an ATPsensitive non-selective cation channel with roles in inflammation and cell death; it is also expressed in osteoblasts where it contributes to normal bone formation. We hypothesized that P2X7R may have a role in the pathogenesis of VC and sought to test this using an in vitro model. METHODS: Primary human coronary artery vascular smooth muscle cells (VSMCs; TCS Cellworks) from two different donors (an 18 year old male and a 58 year old male) at 50-60% confluence were grown for 7 days in normal medium ([Ca2+] 1.8mM, [PO42-] 1.0 mM) or calcification medium containing a high calcium and phosphate concentration ([Ca 2+] 2.7mM, [PO42-] 2.0 mM), with or without lipopolysaccharide (LPS; 10μg/ml) as an inflammatory stimulus, in the absence or presence of the selective P2X7R inhibitor A438079 (1, 3, 10, 20μM; Tocris Bioscience). Calcification was assessed qualitatively by phase contrast microscopy. At day 7, calcification was quantified colorimetrically by the o-cresolphthalein complexone method and corrected for cell protein concentration. Expression of P2X7R and the inflammatory cytokine, interleukin (IL)-1β mRNA was measured by quantitative RT-PCR. RESULTS: High calcium and phosphate concentration induced calcification in the cells of both donors compared to unsupplemented media (18 year old donor: 0.348 μg calcium/μg protein vs 0.016; p<0.001; 58 year old: 1.615 vs 0.072; p<0.001) with more abundant calcification seen in the older donor cells (p<0.001). No significant augmentation in calcification was seen with LPS in cells of either donor. Addition of A438079 (10-20μM) led to a marked reduction in calcification in the 18 year old donor cells (0.085 vs 0.348; p=0.002), also in the 58 year old donor cells (0.987 vs 1.615; p=0.02). These effects were sustained in the presence of LPS. Both LPS and calcification medium induced expression of IL-1β mRNA; A438079 had no effect on expression of this inflammatory cytokine. No significant change in P2X7R mRNA expression was demonstrated with a high calcium and phosphate medium, LPS, or both. CONCLUSION: The P2X7R inhibitor A438079 has a significant abrogating effect on calcification in this in vitro model when tested on primary cells from two donors of different ages, suggesting P2X7R may have an important role in the pathogenesis of VC. Suppression of calcification was not accompanied by a significant change in IL-1β, suggesting the role of P2X7R role in VC may be independent of effects on inflammation. ATP release is frequently coupled to mechanical stretch, so the biological effect of P2X7R inhibition may be underestimated in this non-pulsatile model of arterial VC. Thus, P2X7R seems to be a promising candidate for investigation as a future treatment target for VC in CKD.