Unexpected coordination of copper(II) to a tertiary amide in a
advertisement

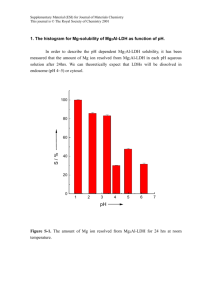
Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 Supplementary Information Synthesis of 4 To macrocyclic amide 2S1 (170 mg, 0.672 mmol) was added ethanol (2 cm3) and the solution buffered to pH 9 (using Merck VWR 9.2 buffer solution, 2 cm3) and CuCl2 (89.5 mg, 0.665 mol) added. The reaction mixture was then heated at reflux for 18 h. The solvent was removed under reduced pressure and the resultant blue solid recrystallised from hot ethanol to yield 4 as blue crystals suitable for single crystal X-ray diffraction (84.4 mg, 33%); m.p. 172-174oC; max/cm-1 1664 (C=O); max/nm 271 (6348), 360 (1215), 580 (105), 714 (120); m/z (ES) 743.4 (4%, 2M-Cl+), 353.1 (50, M-Cl+); (HRMS Found: (M-Cl+) 353.1293 C14H29ClCuN3O requires 353.1290). X-Ray Crystallography The intensity data were collected using an Enraf-Nonius Kappa CCD area detector on an Enraf Nonius FR591 rotating anode generator at 120 (2) K. Graphite monochromated MoK radiation ( = 0.71073 Å) was used with and to fill the Ewald sphere. Data collection, cell refinement and data reduction were carried out using the DENZOS2 and COLLECTS3 packages. Preliminary absorption correction was carried out by multiple scans using SORTAV.S4 The structures were solved by the heavy-atom method using the programs SHELXS-97,S5 and DIRDIFS6 and refined anisotropically (non-hydrogen atoms) by full-matrix least-squares on F2 using SHELXL-97.S5 The H-atom positions were calculated geometrically and refined with a riding model. Data were corrected for absorption using an isotropic non-hydrogen model with the programme XABS2.S7 The program ORTEP-3,S8 was used for graphical representations and WINGXS9 was used to prepare material for publication for all structures. Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 0.7 0.6 0.5 Absorbance 0.4 0 20 40 60 80 100 0.3 0.2 0.1 0 180 280 380 480 580 680 780 880 980 1080 1180 -0.1 -0.2 Wavelength (nm) Fig. S1 UV-visible spectra of 4 in ethanol at room temperature recorded after various times (given in legend in minutes) Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 3 2.5 0 10 25 35 45 65 85 105 165 185 215 245 275 335 395 12h 24h Absorbance 2 1.5 1 0.5 0 180 280 380 480 580 680 780 880 980 1080 1180 -0.5 Wavelength (nm) Fig. S2 Time dependent UV-visible spectra of 4 after refluxing in ethanol (0-395 mins, 12 h and 24 h) Cyclic Voltammetry Cyclic voltammetry was performed using an EG and G Princeton Applied Research Versastat II with Pt working and counter electrodes and an Ag reference electrode. All data were collected in dry degassed DCM solutions with nBu4NCl as the supporting electrolyte using EG and G Model 270/250 Research Electrochemistry Software 4.40. All potentials are referenced relative to Fc/Fc+ at E0 = 0 V. Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 E (mV) -2100 -1900 -1700 -1500 -1300 -1100 -900 -700 -500 200 mV/s 100 mV/s 50 mV/s 25 mV/s Fig. S3 Cyclic Voltammagrams of cathodic sweeps of 4 in DCM with nBu4NCl as the supporting electrolyte referenced to Fc/Fc+. Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 E (mV) -2000 -1800 -1600 -1400 -1200 -1000 -800 -600 15 mV/s 10 mV/s Fig S4 Cyclic votammetry in DCM with nBu4NCl as supporting electrolyte at slow scan rates EPR Spectroscopy X-band spectra (ca. 9.4 GHz) were measured on an EMX hybrid spectrometer and K-band (ca. 24.0 GHz) Qband (ca. 34.0 GHz) spectra on an ESP300 spectrometer. The magnetic fields were calibrated with a Bruker ER035M Gaussmeter and the microwave frequencies measured with an EIP588C microwave counter. Modulation frequencies of 100 KHz were used. EPR simulations were performed using in-house software.S10 From the value of D (ZFS), it is possible to calculate the distance between the two copper atoms, R, using the perturbation theory by Boyd et al.S11 However it is necessary to assume that D is due entirely to dipole-dipole interaction between the two copper(II) atoms and the symmetry arrangements of the two metals are limited where the two dipoles are identical sites of orthorhombic symmetry and the g and D tensor principal axes are all parallel. Considering these limitations, it has been possible to calculate the range of distances R, from the Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 expression of diagonal elements of D tensor,S10 where l, m, n are the direction cosines between the principal axes of the g tensors. z Dxx = gxx2e2(1-3l2)/R3 Dyy = gyy2e2(1-3m2)/R3 Dzz = gzz2e2(1-3n2)/R3 z R n Orientation of the g tensors principal axes. y x m x l y The medium value calculated is 4.8 Å (the values are between 3.9 and 5.9 Å), which is in agreement with the small value of D, suggesting that in frozen solutions of 4 the two copper centres are ligand-bridged, the more likely candidate is chloride. Date: 14.08.1903 Time: 15:22 Filename: C:\MYDOCU~1\JOANNA\WATKIN~1\1463_q1.spc [*10^ 3] 10 5 0 -5 -10 -15 -20 -25 10000 10500 11000 11500 12000 [G] Fig. S5 Powder Q-band (34.0076 GHz) EPR spectrum of 4 at 293 K 12500 13000 Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 Fig. S6 Fluid solution X-band (9.4471 GHz) EPR spectrum of 4 at 293 K in CH2Cl2/toluene 2250 2500 2750 3000 3250 [G] 3500 3750 4000 Fig.S7a X-band (9.4552 GHz) EPR spectrum of 4 and simulation (---) Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 Fig. S7b K-band (24.1795 GHz) EPR spectrum of 4 and simulation (---) Supplementary Material for Dalton Transactions This journal is © The Royal Society of Chemistry 2004 Supplementary References S1 J.E.W. Scheuermann, F. Ronketti, M. Motevalli, D.V. Griffiths and M. Watkinson, New J. Chem., 2002, 26, 1054. S2 DENZO Data collection and processing software: Z. Otwinowski and W. Minor, Methods in Enzymology, Volume 276: Macromolecular Crystallography, part A, p.307-326, 1997, C.W. Carter, Jr and R.M. Sweet, Eds., Academic Press. S3 COLLECT Data collection and processing user interface: Collect: Data collection software, R. Hooft, Nonius B.V., 1998. S4 SORTAV absorption correction software package: R.H. Blessing, Acta Crystallogr., Sect. A., 1995, 51, 33 and R.H. Blessing, J. Appl. Cryst., 1997, 30, 421. S5 G.M. Sheldrick, 1997 SHELX-97. Program for solution and Refinement of Crystal Structures. University of Göttingen, Germany. S6 DIRDIF-99 program system. P.T. Beurskens, G. Beurskens, W.P. Bosman, R. de Gelder, S. GarciaGranda, R.O. Gould, R. Israel and J.M.M. Smits, Crystallography Laboratory, University of Nijmegen, The Netherlands. 1999. S7 S. Parkin, B. Moezzi and H. Hope, J. Appl. Cryst., 1995, 28, 53. S8 L.J. Farrugia, ORTEP-3 for Windows. J. Appl. Crystallogr., 1997, 30, 565. S9 L.J. Farrugia, WinGX- A Windows Program for Crystal Structure Analysis, University of Glasgow, Glasgow, 1998. S10 F.E. Mabbs and D. Collison, Electron Paramagnetic Resonance of d Transition Metal Compound, Elsevier, Amsterdam, 1992. S11 P.D.W. Boyd, A.D. Toy and T.D. Smith, J. Chem. Soc., Dalton Trans., 1973, 1549.