Synthesis of mppe, discussion and additional figures
advertisement

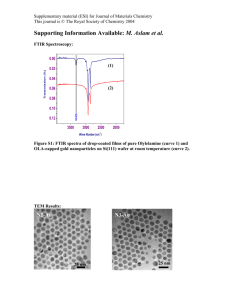
Supplementary Material for Chemical Communications This journal is © The Royal Society of Chemistry 2003 Supporting information for Coordination polymers based on square planar Co(II) node and linear spacer: solvent-dependent pseudo-polymorphism and an unprecedented interpenetrating structure containing both 2D and 3D topological isomers Dong Mok Shin,a In Su Lee, b Young Keun Chung* a and Myoung Soo Lahc a Department of Chemistry and the Center for Molecular Catalysis, College of Natural Sciences, Seoul National University, Seoul 151-742, Korea b Advanced Materials Research Institute, LG Chemical Ltd. Research Park, Taejon, 305- 380, Korea c Department of Applied Chemistry and Chemistry, College of Science and Technology, Han Yang Unversity, Ansan 425-170, Korea Synthesis of mppe. To a solution of LDA (generated in situ by the reaction of diisopropylamine (1.8 ml, 12.8 mmol) in 30 ml of THF with n-BuLi (5.5 ml, 13.8 mmol) at -78 °C) was added 4-methyl pyrimidine (1.0 ml, 11 mmol) at -78°C. While the solution was stirred at room temperature for 30 min, the solution turned to yellowish. To the yellowish solution was added 4-acetyl pyridine (1.0 ml, 9.0 mmol) at room temperature. The resulting solution was stirred for 4 h and quenched by addition of water (30 ml) and methylene dichloride (30 ml). The methylene dichloride layer was collected, evaporated to dryness, and dissolved in 15 ml of pyridine. The pyridine solution was cooled to 0°C. Excess POCl3 (2 ml) was added dropwise to the pyridine solution. The resulting solution was stirred at room temperature for 3h and quenched by Supplementary Material for Chemical Communications This journal is © The Royal Society of Chemistry 2003 addition of ice. After evaporation of pyridine, the residue was dissolved in water (30 ml) and basified by addition of aqueous 4 M NaOH. Extraction with dichloromethane (100 ml) followed by chromatography on a silica gel column eluting with Et2O/MeOH (v/v, 10:1) gave 0.50 g of 5 (28 %). 1H NMR (CDCl3) 9.26 (s 1 H), 8.73 (d, 5.2 Hz, 1 H), 8.65 (d, 4.3 Hz, 2 H), 7.42 (2, 4.6 Hz, 2 H), 7.29 (d, 5.2 Hz, 1 H), 6.81 (s, 1 H) 2.62 (s, 3 H) ppm; 13 C NMR (CDCl3) 163.36, 158.95, 157.57, 150.88, 150.54, 127.09, 122.06, 121.13, 18.00; HRMS M+ calc. 197.0953, obsd. 197.0954. Supplementary Material for Chemical Communications This journal is © The Royal Society of Chemistry 2003 Discussion about the color change of 2 with/without the solvent: The color change to blue might suggest desorption of the solvent followed by a concomitant structural change from octahedral to tetrahedral. Usually this kind of change may result in an overall change in the crystal. However, in our case, we could not observe any changes in the XRD pattern before and after desorption of the solvent. Thus, we imagine that a certain amount of cobalt metals are converted to a tetrahedral geometry and a mixture of tetrahedral and octahedral cobalt ions coexists in the blue crystal. If we assume that the change occurs in a small portion of the cobalt ions or a newly formed phase is amorphous, the domain formed is not large enough to be detected by the change in an XRD pattern. In these circumstances, we could only observe the pre-existing crystalline phase, octahedral cobalt metals ions, even though we could see only blue crystals by eye. A quite similar case was reported by Kitagawa et. al. in Chem. Eur. J. 2002, 8, 3587. We also carried out TGA and DSC study. However, we could not see any evidence for a special phase change or decomposition. We also did an IR study in order to confirm whether the SCN ligand acts as a bridging ligand. However, we did not find out any special indication. (a) (b) Figure 1. Color change of the crystal 2. (a) Right after taking out the crystal. (b) 1 hr later. Supplementary Material for Chemical Communications This journal is © The Royal Society of Chemistry 2003 X R P D s p e c tra o f 2 (a ) (b ) 5 10 15 20 25 30 35 40 45 2 th e ta Figure 2. Powder XRD spectra of 2. (a) in the solvent. (b) after 2days drying in 80oC . 50 Supplementary Material for Chemical Communications This journal is © The Royal Society of Chemistry 2003 TGA of 2 3.5 3 weight 2.5 2 1.5 Solvent loosing region 1 0.5 0 0 50 100 150 200 250 300 350 400 450 o Temp( C) (a) D S C of 2 1.5 1 0.5 0 0 20 40 60 80 100 -0.5 -1 -1.5 Tem p(o C ) (b) Figure 3. (a) TGA of 2. (b) DSC of 2. 120 140 160 180 Supplementary Material for Chemical Communications This journal is © The Royal Society of Chemistry 2003 Figure 4. IR spectrum of 2. CN stretching of SCN ligand at 2064 cm-1.